Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
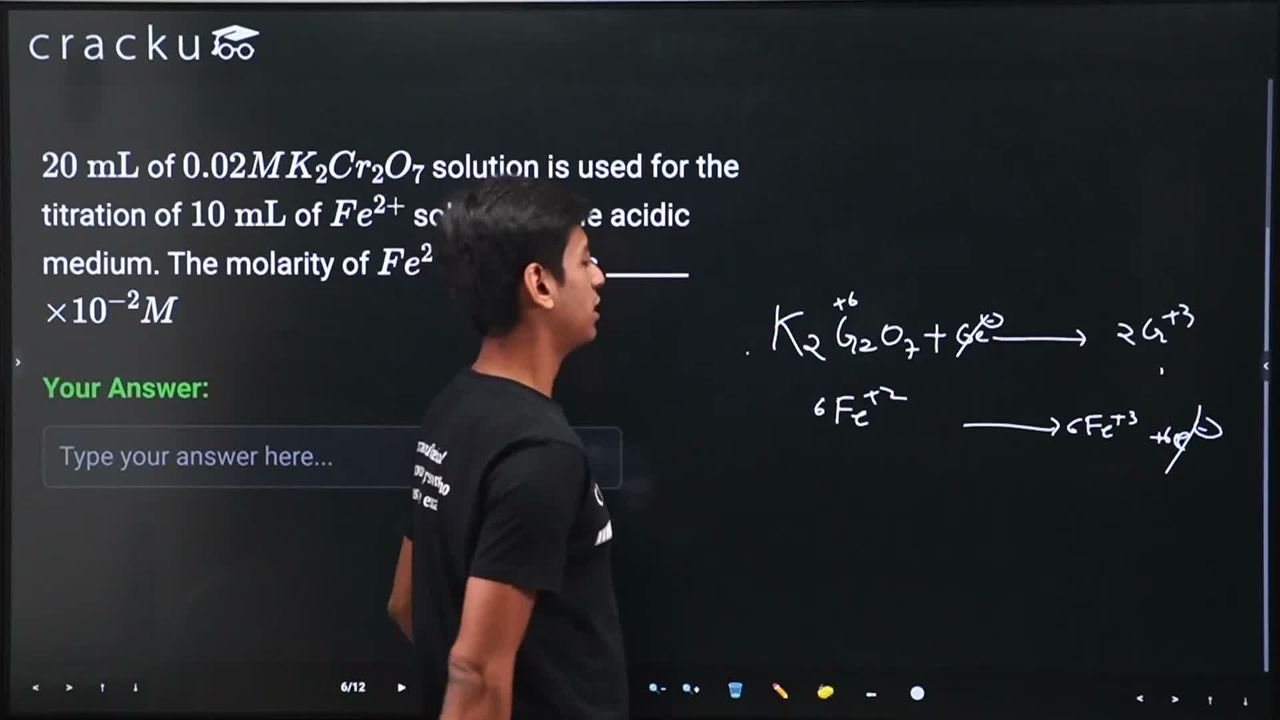
$$20 \text{ mL}$$ of $$0.02M K_2Cr_2O_7$$ solution is used for the titration of $$10 \text{ mL}$$ of $$Fe^{2+}$$ solution in the acidic medium. The molarity of $$Fe^{2+}$$ solution is ______ $$\times 10^{-2} M$$
Correct Answer: 24
We are given that 20 mL of 0.02 M $$K_2Cr_2O_7$$ is used to titrate 10 mL of $$Fe^{2+}$$ solution in acidic medium.
The balanced redox reaction in acidic medium is:
$$K_2Cr_2O_7 + 6Fe^{2+} + 14H^+ \rightarrow 2Cr^{3+} + 6Fe^{3+} + 2K^+ + 7H_2O$$
From the stoichiometry, 1 mole of $$K_2Cr_2O_7$$ reacts with 6 moles of $$Fe^{2+}$$.
Moles of $$K_2Cr_2O_7$$ used:
$$n_{K_2Cr_2O_7} = M \times V = 0.02 \times \frac{20}{1000} = 4 \times 10^{-4} \text{ mol}$$
Moles of $$Fe^{2+}$$ that react:
$$n_{Fe^{2+}} = 6 \times n_{K_2Cr_2O_7} = 6 \times 4 \times 10^{-4} = 24 \times 10^{-4} \text{ mol}$$
Molarity of $$Fe^{2+}$$ solution:
$$M_{Fe^{2+}} = \frac{n_{Fe^{2+}}}{V} = \frac{24 \times 10^{-4}}{10/1000} = \frac{24 \times 10^{-4}}{10^{-2}} = 24 \times 10^{-2} \text{ M}$$
Therefore, the answer is $$\textbf{24}$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
