Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The aldehydes which will not form Grignard product with one equivalent of Grignard reagents are:
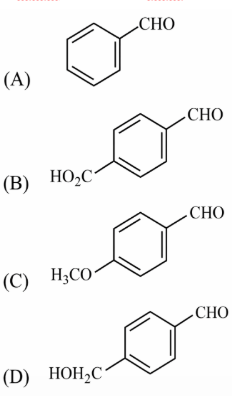
The reaction in question is the addition of a single equivalent of a Grignard reagent, written in the general form $$\mathrm{R{-}MgX},$$ to the carbonyl group of an aldehyde. A necessary condition for this nucleophilic addition is that the Grignard reagent should reach the carbonyl carbon without being destroyed en-route. The reagent is both a very strong base and a very strong nucleophile; therefore any group present in the substrate that is (i) proton-donating (acidic) or (ii) itself an electrophilic carbonyl capable of reacting faster than the aldehyde will consume the Grignard reagent before it can add to the aldehyde carbonyl.
We first recall two key facts.
1. A Grignard reagent instantly abstracts an acidic proton. Symbolically,
$$\mathrm{R{-}MgX \;+\; H{-}Z \;\longrightarrow\; R{-}H \;+\; Z^{-}MgX^{+}},$$
where $$\mathrm{H{-}Z}$$ is any group with an O-H, N-H, S-H, or similarly acidic bond.
2. A Grignard reagent will add to a carbonyl group provided it has not already been destroyed by acid-base reaction.
Now we examine each option.
(A) Benzaldehyde, $$\mathrm{C_6H_5{-}CHO}.$$
No acidic proton other than the aldehydic one, and that proton is attached to carbon, not to a hetero-atom. Hence the Grignard reagent reaches the carbonyl safely and adds:
$$\mathrm{R{-}MgX + C_6H_5{-}CHO \xrightarrow{Et_2O} C_6H_5{-}CH(OMgX)R \xrightarrow{H_2O} C_6H_5{-}CH(OH)R}$$
So benzaldehyde does give the expected Grignard product.
(B) An aldehyde that also contains a $$\mathrm{HO_2C{-}}$$ (carboxylic acid) group.
The -COOH unit possesses an acidic O-H proton. Using the acid-base equation stated above, one equivalent of the Grignard reagent is consumed immediately:
$$\mathrm{R{-}MgX + HO_2C{-}R' \longrightarrow R{-}H + ^{-}O_2C{-}R' \,MgX^{+}}$$
With the reagent destroyed, nothing is left to attack the aldehyde carbonyl. No addition product appears.
(C) An aldehyde that also contains a $$\mathrm{H_3CO{-}}$$ (methoxy) group.
The methoxy group has no acidic proton; it is an ether. Therefore the Grignard reagent is not quenched and can still add to the aldehyde carbonyl exactly as in (A). Product formation is unimpeded.
(D) An aldehyde that also contains a $$\mathrm{HOCH_2{-}}$$ (alcohol, -CH2OH) group.
The alcohol O-H is acidic. One equivalent of Grignard reagent is again lost through proton abstraction:
$$\mathrm{R{-}MgX + HOCH_2{-}R' \longrightarrow R{-}H + ^{-}OCH_2{-}R' \,MgX^{+}}$$
Once more, no nucleophile remains to attack the aldehydic carbonyl, and no Grignard addition product is obtained.
Summarising, aldehydes (B) and (D) fail to furnish Grignard addition products with one equivalent of Grignard reagent, while (A) and (C) succeed.
Hence, the correct answer is Option D.
Create a FREE account and get:
Educational materials for JEE preparation
