Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
One mole of an ideal monoatomic gas undergoes two reversible processes (A $$\to$$ B and B $$\to$$ C) as shown in the given figure:
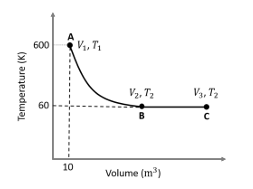
A $$\to$$ B is an adiabatic process. If the total heat absorbed in the entire process (A $$\to$$ B and B $$\to$$ C) is $$RT_2 \ln 10$$, the value of $$2 \log V_3$$ is ________.
[Use, molar heat capacity of the gas at constant pressure, $$C_{p,m} = \frac{5}{2}R$$]
Correct Answer: 7
Create a FREE account and get:
Educational materials for JEE preparation
