Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In a one-litre flask, 6 moles of A undergoes the reaction A(g) $$\rightleftharpoons$$ P(g). The progress of product formation at two temperatures (in Kelvin), $$T_1$$ and $$T_2$$, is shown in the figure:
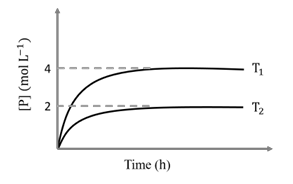
If $$T_1 = 2T_2$$ and $$(\Delta G_2^\circ - \Delta G_1^\circ) = RT_2 \ln x$$, then the value of x is ______.
[$$\Delta G_1^\circ$$ and $$\Delta G_2^\circ$$ are standard Gibb's free energy change for the reaction at temperatures $$T_1$$ and $$T_2$$, respectively.]
Correct Answer: 8
The flask volume is 1 L, hence the numerical values of moles and molarities are the same.
Initial state (common to both runs):
number of moles of $$A = 6$$,
number of moles of $$P = 0$$.
From the graph (plateau values):
at temperature $$T_1$$, moles of $$P$$ formed $$=4$$ ⇒ moles of $$A$$ left $$=6-4=2$$;
at temperature $$T_2$$, moles of $$P$$ formed $$=2$$ ⇒ moles of $$A$$ left $$=6-2=4$$.
The reaction is $$A(g) \rightleftharpoons P(g)$$, so
$$K = \frac{[P]}{[A]}$$.
Equilibrium constants:
$$K_1 = \frac{4}{2} = 2$$ at $$T_1$$;
$$K_2 = \frac{2}{4} = 0.5$$ at $$T_2$$.
Standard Gibbs free energy relation:
$$\Delta G^\circ = -RT \ln K$$.
Hence
$$\Delta G_1^\circ = -R T_1 \ln K_1$$,
$$\Delta G_2^\circ = -R T_2 \ln K_2$$.
The required difference is
$$\Delta G_2^\circ - \Delta G_1^\circ = -R T_2 \ln K_2 + R T_1 \ln K_1$$.
Given $$T_1 = 2T_2$$, substitute:
$$\Delta G_2^\circ - \Delta G_1^\circ = R T_2 \left(2\ln K_1 - \ln K_2\right)$$.
Combine the logarithms:
$$2\ln K_1 - \ln K_2 = \ln \left(\frac{K_1^2}{K_2}\right)$$.
Therefore
$$\Delta G_2^\circ - \Delta G_1^\circ = R T_2 \ln\!\left(\frac{K_1^2}{K_2}\right).$$
Comparing with $$\Delta G_2^\circ - \Delta G_1^\circ = R T_2 \ln x$$ gives
$$x = \frac{K_1^2}{K_2} = \frac{(2)^2}{0.5} = \frac{4}{0.5} = 8.$$
Hence, the value of $$x$$ is 8.
Create a FREE account and get:
Educational materials for JEE preparation
