Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The plot of log $$k_f$$ versus $$1/T$$ for a reversible reaction A(g) $$\rightleftharpoons$$ P(g) is shown.
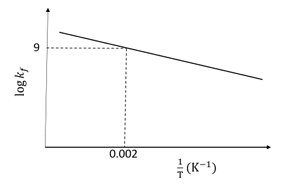
Pre-exponential factors for the forward and backward reactions are $$10^{15}$$ s$$^{-1}$$ and $$10^{11}$$ s$$^{-1}$$, respectively. If the value of log K for the reaction at 500 K is 6, the value of $$|\log k_b|$$ at 250 K is ______.
[K = equilibrium constant of the reaction,
$$k_f$$ = rate constant of forward reaction,
$$k_b$$ = rate constant of backward reaction]
Correct Answer: 5
Create a FREE account and get:
Educational materials for JEE preparation
