NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 31
A sample of CaCO$$_3$$ and MgCO$$_3$$ weighed 2.21 g is ignited to constant weight of 1.152 g. The composition of the mixture is: (Given molar mass in g mol$$^{-1}$$, CaCO$$_3$$: 100, MgCO$$_3$$: 84)
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 32
The four quantum numbers for the electron in the outer most orbital of potassium (atomic no. 19) are
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 33
Consider the following elements.
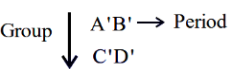
Which of the following is/are true about A', B', C' and D'?
A. Order of atomic radii: $$B' < A' < D' < C'$$
B. Order of metallic character: $$B' < A' < D' < C'$$
C. Size of the element: $$D' < C' < B' < A'$$
D. Order of ionic radii: $$B'^{+} < A'^{+} < D'^{+} < C'^{+}$$
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 34
Which of the following is least ionic?
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 35
$$A_g \rightleftharpoons B_g + \frac{C}{2}_g$$. The correct relationship between $$K_P$$, $$\alpha$$ and equilibrium pressure $$P$$ is
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 36
Given below are two statements:
Statement I: $$S_8$$ solid undergoes disproportionation reaction under alkaline conditions to form $$S^{2-}$$ and $$S_2O_3^{2-}$$
Statement II: $$ClO_4^{-}$$ can undergo disproportionation reaction under acidic condition.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 37
Given below are two statements:
Statement I: Group 13 trivalent halides get easily hydrolysed by water due to their covalent nature.
Statement II: AlCl$$_3$$ upon hydrolysis in acidified aqueous solution forms octahedral $$[Al(H_2O)_6]^{3+}$$ ion.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 38
Identify structure of 2, 3-dibromo-1-phenylpentane.
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 39
The fragrance of flowers is due to the presence of some steam volatile organic compounds called essential oils. These are generally insoluble in water at room temperature but are miscible with water vapour in the vapour phase. A suitable method for the extraction of these oils from the flowers is:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 40
Major product of the following reaction is:
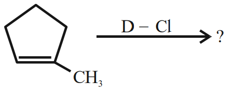
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 41
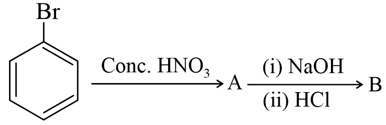
Identify $$A$$ and $$B$$ in the following reaction sequence:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 42
Choose the correct statements from the following:
A. All group 16 elements form oxides of general formula $$EO_2$$ and $$EO_3$$ where $$E = S, Se, Te$$ and $$Po$$. Both the types of oxides are acidic in nature.
B. $$TeO_2$$ is an oxidising agent while $$SO_2$$ is reducing in nature.
C. The reducing property decreases from $$H_2S$$ to $$H_2Te$$ down the group.
D. The ozone molecule contains five lone pairs of electrons.
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 43
Choose the correct statements from the following:
A. $$Mn_2O_7$$ is an oil at room temperature
B. $$V_2O_4$$ reacts with acid to give $$VO_2^{2+}$$
C. CrO is a basic oxide
D. $$V_2O_5$$ does not react with acid
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 44
Select the option with correct property:
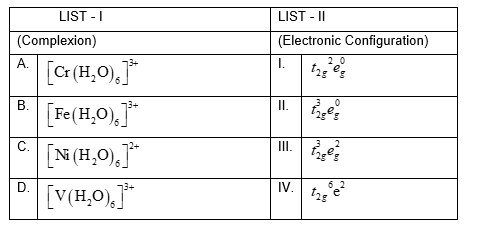
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 45
Match List I with List II
Choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 46
The correct order of reactivity in electrophilic substitution reaction of the following compounds is:
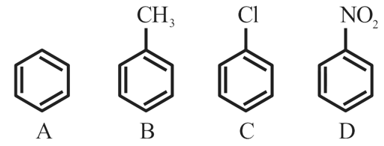
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 47
Identify the name reaction:
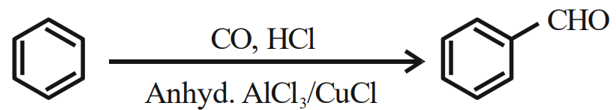
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 48
Identify major product 'P' formed in the following reaction:
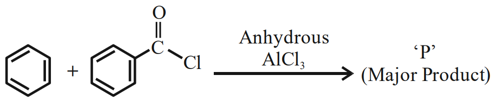
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 49
The azo-dye $$Y$$ formed in the following reactions is: Sulphanilic acid + NaNO$$_2$$ + CH$$_3$$COOH $$\rightarrow$$ X
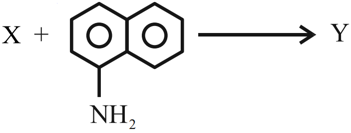
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 50
Given below are two statements:
Statement I: Aniline reacts with con. $$H_2SO_4$$ followed by heating at 453-473 K gives p-aminobenzene sulphonic acid, which gives blood red colour in the 'Lassaigne's test.
Statement II: In Friedel-Craft's alkylation and acylation reactions, aniline forms salt with the $$AlCl_3$$ catalyst. Due to this, nitrogen of aniline acquires a positive charge and acts as deactivating group.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 51
The molarity of 1 L orthophosphoric acid $$H_3PO_4$$ having 70% purity by weight (specific gravity 1.54 g cm$$^{-3}$$) is ______ M. (Molar mass of $$H_3PO_4 = 98$$ g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 52
A diatomic molecule has a dipole moment of 1.2 D. If the bond distance is 1 $$\mathring{A}$$, then fractional charge on each atom is ______ $$\times 10^{-1}$$ esu. (Given 1D = $$10^{-18}$$ esu cm)
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 53
If 5 moles of an ideal gas expands from 10 L to a volume of 100 L at 300 K under isothermal and reversible condition then work, $$w$$, is $$-x$$ J. The value of $$x$$ is (Given $$R = 8.314$$ J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 54
Number of isomeric products formed by monochlorination of 2-methylbutane in presence of sunlight is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 55
The values of conductivity of some materials at 298.15 K in S m$$^{-1}$$ are $$2.1 \times 10^3$$, $$1.0 \times 10^{-16}$$, $$1.2 \times 10$$, $$3.91$$, $$1.5 \times 10^{-2}$$, $$1 \times 10^{-7}$$, $$1.0 \times 10^3$$. The number of conductors among the materials is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 56
$$r = kA$$ for a reaction, 50% of A is decomposed in 120 minutes. The time taken for 90% decomposition of A is ______ minutes.
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 57
Number of moles of $$H^+$$ ions required by 1 mole of $$MnO_4^-$$ to oxidise oxalate ion to $$CO_2$$ is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 58
In the reaction of potassium dichromate, potassium chloride and sulfuric acid (conc.), the oxidation state of the chromium in the product is +
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 59
A compound $$x$$ with molar mass 108 g mol$$^{-1}$$ undergoes acetylation to give product with molar mass 192 g mol$$^{-1}$$. The number of amino groups in the compound $$x$$ is
789
456
123
0.-
Clear All
NTA JEE Mains 31st Jan 2024 Shift 2 - Chemistry - Question 60
From the vitamins $$A, B_1, B_6, B_{12}, C, D, E$$ and $$K$$, the number of vitamins that can be stored in our body is
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




