Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A compound $$x$$ with molar mass 108 g mol$$^{-1}$$ undergoes acetylation to give product with molar mass 192 g mol$$^{-1}$$. The number of amino groups in the compound $$x$$ is
Correct Answer: 2
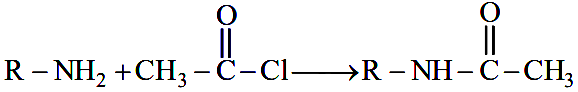
In this reaction, an H atom (mass 1) on the $$-\text{NH}_2$$ group is replaced by an acetyl group $$-\text{COCH}_3$$ (mass 43). The net increase in molar mass per amino group is $$43 - 1 = 42$$ g/mol.
Since the product’s molar mass increases from 108 to 192, the total increase in molar mass is $$\Delta M = 192 - 108 = 84 \, \text{g/mol}$$.
Substituting into the relation for the number of amino groups gives $$n = \frac{\Delta M}{42} = \frac{84}{42} = 2$$.
Therefore, the compound $$X$$ has 2 amino groups, corresponding to Option 2.

Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
