Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
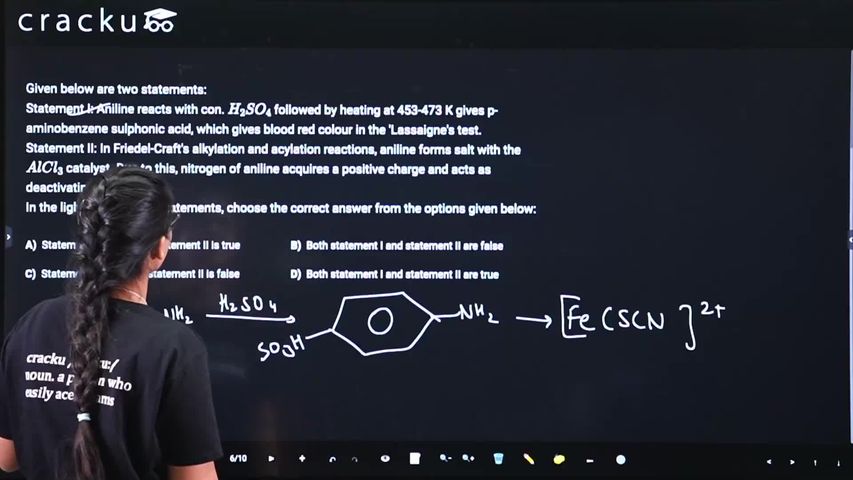
Given below are two statements:
Statement I: Aniline reacts with con. $$H_2SO_4$$ followed by heating at 453-473 K gives p-aminobenzene sulphonic acid, which gives blood red colour in the 'Lassaigne's test.
Statement II: In Friedel-Craft's alkylation and acylation reactions, aniline forms salt with the $$AlCl_3$$ catalyst. Due to this, nitrogen of aniline acquires a positive charge and acts as deactivating group.
In the light of the above statements, choose the correct answer from the options given below:
Statement I deals with the sulfonation of aniline and the Lassaigne’s test for sulfur. Sulfonation of aniline with conc. $$H_2SO_4$$ at 453-473 K proceeds as follows:
$$C_6H_5NH_2 + H_2SO_4 \xrightarrow{453-473\,K} p\text{-}H_2NC_6H_4SO_3H$$
The product is p-aminobenzenesulfonic acid (sulfanilic acid).
In the Lassaigne’s test for sulfur, the sodium fusion extract is treated with sodium nitroprusside reagent. The presence of sulfur gives a blood-red coloration. Since p-aminobenzenesulfonic acid contains a sulfonic acid group (-SO₃H), it yields a blood-red color in this test. Therefore, Statement I is true.
Statement II concerns Friedel-Crafts reactions of aniline. Aniline’s -lone pair forms a complex with the Lewis acid catalyst $$AlCl_3$$:
$$C_6H_5NH_2 + AlCl_3 \longrightarrow [C_6H_5NH_2\cdot AlCl_3] \longrightarrow [C_6H_5NH_3]^+ + AlCl_4^-$$
In this complex, the nitrogen carries a positive charge, withdrawing electron density from the benzene ring and making it less reactive toward electrophilic substitution. Hence aniline acts as a deactivating group under Friedel-Crafts conditions, and no alkylation or acylation occurs. Therefore, Statement II is true.
Both Statement I and Statement II are true. The correct answer is Option D.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
