NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 31
Match List I with List II
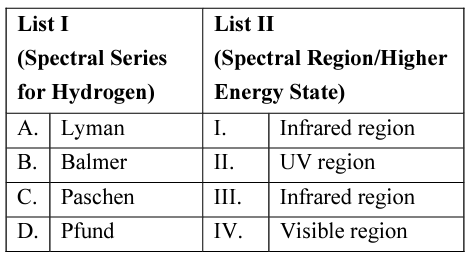
Choose the correct answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 32
The element having the highest first ionization enthalpy is
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 33
Given below are two statements:
Statement I: Fluorine has most negative electron gain enthalpy in its group.
Statement II: Oxygen has least negative electron gain enthalpy in its group.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 34
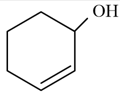
According to IUPAC system, the compound is named as:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 35
The ascending acidity order of the following H atoms is
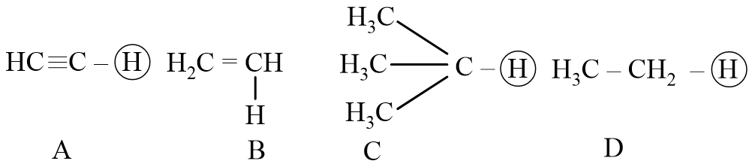
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 36
Match List I with List II
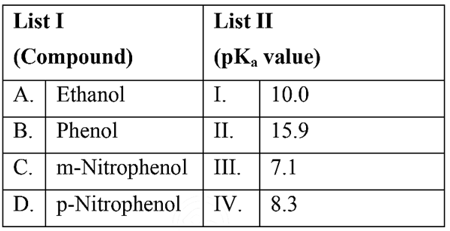
Choose the correct answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 37
Which one of the following will show geometrical isomerism?
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 38
Chromatographic technique/s based on the principle of differential adsorption is/are
A. Column chromatography B. Thin layer chromatography C. Paper chromatography
Choose the most appropriate answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 39
Anomalous behaviour of oxygen is due to its
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 40
Which of the following acts as a strong reducing agent? (Atomic number: Ce = 58, Eu = 63, Gd = 64, Lu = 71)
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 41
Which of the following statements are correct about Zn, Cd and Hg?
A. They exhibit high enthalpy of atomization as the d-subshell is full.
B. Zn and Cd do not show variable oxidation state while Hg shows +I and +II.
C. Compounds of Zn, Cd and Hg are paramagnetic in nature.
D. Zn, Cd and Hg are called soft metals.
Choose the most appropriate from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 42
The correct IUPAC name of $$K_2MnO_4$$ is:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 43
Alkyl halide is converted into alkyl isocyanide by reaction with
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 44
Phenol treated with chloroform in presence of sodium hydroxide, which further hydrolysed in presence of an acid results
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 45
Identify the reagents used for the following conversion
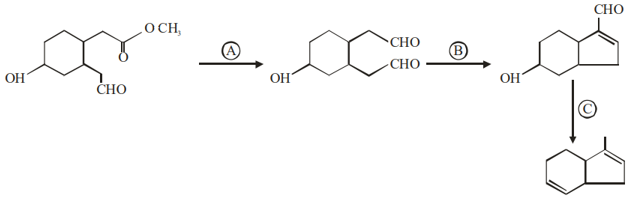
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 46
Which of the following reaction is correct?
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 47
The product A formed in the following reaction is:
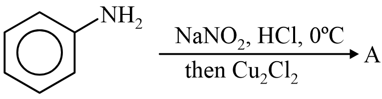
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 48
On passing a gas, '$$X$$', through Nessler's reagent, a brown precipitate is obtained. The gas '$$X$$' is
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 49
A reagent which gives brilliant red precipitate with Nickel ions in basic medium is
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 50
Match List I with List II
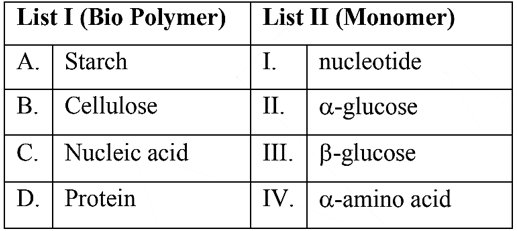
Choose the correct answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 51
The total number of molecules with zero dipole moment among $$CH_4, BF_3, H_2O, HF, NH_3, CO_2$$ and $$SO_2$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 52
The total number of 'Sigma' and Pi bonds in 2-formylhex-4-enoic acid is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 53
The total number of anti bonding molecular orbitals, formed from $$2s$$ and $$2p$$ atomic orbitals in a diatomic molecule is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 54
Standard enthalpy of vapourisation for $$CCl_4$$ is $$30.5$$ kJ mol$$^{-1}$$. Heat required for vapourisation of $$284$$ g of $$CCl_4$$ at constant temperature is ______ kJ. (Given molar mass in g mol$$^{-1}$$; C = 12, Cl = 35.5)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 55
The following concentrations were observed at $$500$$ K for the formation of $$NH_3$$ from $$N_2$$ and $$H_2$$. At equilibrium: $$[N_2] = 2 \times 10^{-2}$$ M, $$[H_2] = 3 \times 10^{-2}$$ M and $$[NH_3] = 1.5 \times 10^{-2}$$ M. Equilibrium constant for the reaction is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 56
If $$50$$ mL of $$0.5$$ M oxalic acid is required to neutralise $$25$$ mL of NaOH solution, the amount of NaOH in $$50$$ mL of given NaOH solution is ______ g.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 57
Molality of $$0.8$$ M $$H_2SO_4$$ solution (density $$1.06$$ g cm$$^{-3}$$) is ______ $$\times 10^{-3}$$ m.
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 58
A constant current was passed through a solution of $$AuCl_4^-$$ ion between gold electrodes. After a period of $$10.0$$ minutes, the increase in mass of cathode was $$1.314$$ g. The total charge passed through the solution is ______ $$\times 10^{-2}$$ F. (Given atomic mass of Au = 197)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 59
The half-life of radioisotopic bromine-82 is $$36$$ hours. The fraction which remains after one day is ______ $$\times 10^{-2}$$.
(Given antilog $$0.2006 = 1.587$$)
789
456
123
0.-
Clear All
NTA JEE Mains 29th Jan 2024 Shift 2 - Chemistry - Question 60
Oxidation state of Fe (Iron) in complex formed in Brown ring test.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




