Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
We need to determine which element among Si, Al, N, and C has the highest first ionization enthalpy.
The first ionization enthalpies (approximate values) are:
Al (Z = 13): 577 kJ/mol
Si (Z = 14): 786 kJ/mol
C (Z = 6): 1086 kJ/mol
N (Z = 7): 1402 kJ/mol
General trend: Ionization enthalpy increases across a period from left to right. However, there are exceptions due to electronic configuration stability.
Why Nitrogen has the highest IE among these elements:
Nitrogen has the electronic configuration $$1s^2\, 2s^2\, 2p^3$$. The $$2p$$ subshell is exactly half-filled, with one electron in each of the three $$2p$$ orbitals. This half-filled configuration provides extra stability due to:
1. Maximum exchange energy
2. Symmetrical distribution of electrons
This extra stability makes it significantly harder to remove an electron from nitrogen compared to the other elements listed.
The correct answer is Option 3: N (Nitrogen).
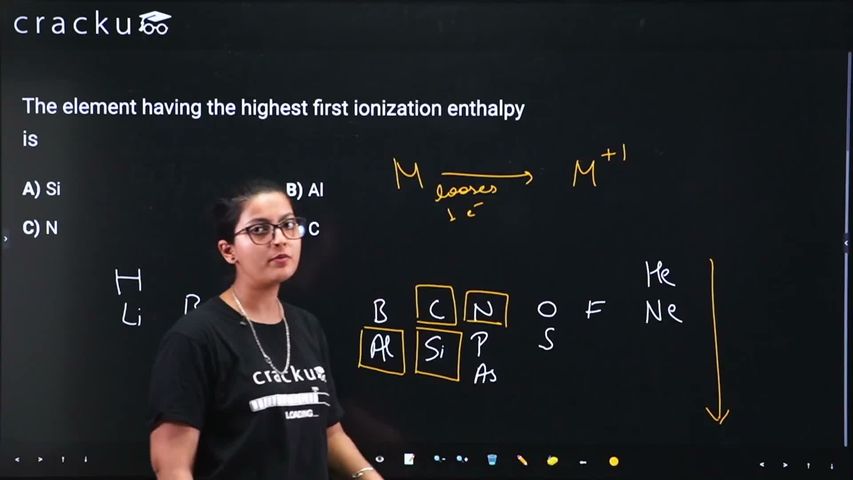
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
