NTA JEE Mains 24th january 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 31
It is observed that characteristic X-ray spectra of elements show regularity. When frequency to the power 'n' i.e. $$\nu^n$$ of X-rays emitted is plotted against atomic number Z, the following graph is obtained.
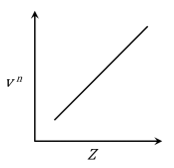
The value of 'n' is
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 32
Decreasing order of the hydrogen bonding in following forms of water is correctly represented by
A. Liquid water
B. Ice
C. Impure water
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 33
Order of Covalent bond;
A.$$KF > KI; LiF > KF$$
B. $$KF < KI; LiF > KF$$
C. $$SnCl_4 > SnCl_2$$; CuCl > NaCl
D. $$LiF > KF; CuCl > NaCl$$
E. $$KF < KI; CuCl > NaCl$$
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 34
Match Column I with Column II

NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 35
Reaction of BeO with ammonia and hydrogen fluoride gives 'A' which on thermal decomposition gives $$BeF_2$$ and $$NH_4F$$. What is 'A' ?
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 36
Increasing order of stability of the resonance structure is:
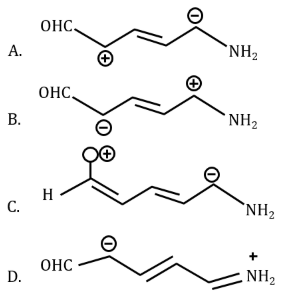
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 37
Which of the following is true about freons?
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 38
In the depression of freezing point experiment
A. Vapour pressure of the solution is less than that of pure solvent
B. Vapour pressure of the solution is more than that of pure solvent
C. Only solute molecules solidify at the freezing point
D. Only solvent molecules solidify at the freezing point
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 39
Statement I: For colloidal particles, the values of colligative properties are of small order as compared to values shown by true solutions at same concentration.
Statement II: For colloidal particles, the potential difference between the fixed layer and the diffused layer of same charges is called the electrokinetic potential or zeta potential.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 40
Match List I with List II.

NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 41
An ammoniacal metal salt solution gives a brilliant red precipitate on addition of dimethylglyoxime. The metal ion is :
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 42
Which of the Phosphorus oxoacid can create silver mirror from $$AgNO_3$$ solution?
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 43
The magnetic moment of a transition metal compound has been calculated to be 3.87 BM. The metal ion is
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 44
The primary and secondary valencies of cobalt respectively in $$Co(NH_3)_5ClCl_2$$ are:
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 45
Assertion A: Hydrolysis of an alkyl chloride is a slow reaction but in the presence of NaI, the rate of hydrolysis increases.
Reason R: $$I^-$$ is a good nucleophile as well as a good leaving group.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 46
In the following given reaction 'A' is:
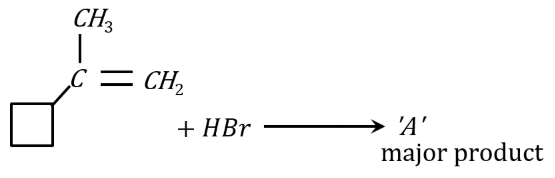
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 47
'A' and 'B' formed in the following set of reactions are:
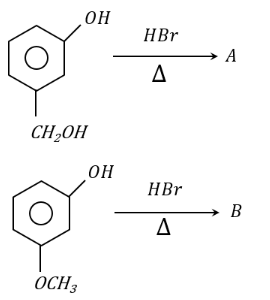
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 48
'R' formed in the following sequence of reaction is:
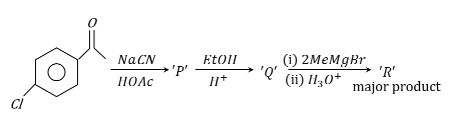
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 49
Compound X undergoes following sequence of reactions to give the Lactone Y.
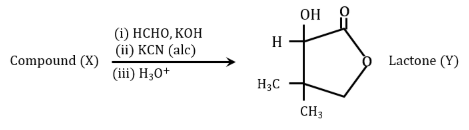
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 50
Given below are two statements :
Statement I : Noradrenaline is a neurotransmitter.
Statement II : Low level of noradrenaline is not the cause of depression in human.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 51
If wavelength of the first line of the Paschen series of hydrogen atom is 720 nm, then the wavelength of the second line of this series is _______ nm. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 52
For independent process at 300 K.

The number of non-spontaneous process from the following is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 53
The dissociation constant of acetic acid is $$x \times 10^{-5}$$. When 25 mL of 0.2 M $$CH_3COONa$$ solution is mixed with 25 mL of 0.02 M $$CH_3COOH$$ solution, the pH of the resultant solution is found to be equal to 5. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 54
Uracil is base present in RNA with the following structure. % of N in uracil is _____.
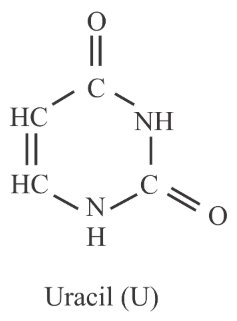
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 55
When $$Fe_{0.93}O$$ is heated in presence of oxygen, it converts to $$Fe_2O_3$$. The number of correct statement/s from the following is ______.
A. The equivalent weight of $$Fe_{0.93}O$$ is $$\frac{\text{Molecular weight}}{0.79}$$
B. The number of moles of $$Fe^{2+}$$ and $$Fe^{3+}$$ in 1 mole of $$Fe_{0.93}O$$ is 0.79 and 0.14 respectively.
C. $$Fe_{0.93}O$$ is metal deficient with lattice comprising of cubic closed packed arrangement of $$O^{2-}$$ ions.
D. The % composition of $$Fe^{2+}$$ and $$Fe^{3+}$$ in $$Fe_{0.93}O$$ is 85% and 15% respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 56
5 g of NaOH was dissolved in deionized water to prepare a 450 mL stock solution. What volume (in mL) of this solution would be required to prepare 500 mL of 0.1 M solution? Given : Molar Mass of Na, O and H is 23, 16 and 1 g mol$$^{-1}$$ respectively
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 57
At 298 K, a 1 litre solution containing 10 mmol of $$Cr_2O_7^{2-}$$ and 100 mmol of $$Cr^{3+}$$ shows a pH of 3.0. Given : $$Cr_2O_7^{2-} \to Cr^{3+}$$; $$E^0 = 1.330$$ V and $$\frac{2.303RT}{F} = 0.059$$ V. The potential for the half cell reaction is $$x \times 10^{-3}$$ V. The value of x is _____.
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 58
The number of correct statement/s from the following is _____.
A. Larger the activation energy, smaller is the value of the rate constant.
B. The higher is the activation energy, higher is the value of the temperature coefficient.
C. At lower temperatures, increase in temperature causes more change in the value of k than at higher temperature.
D. A plot of $$\ln k$$ vs $$\frac{1}{T}$$ is a straight line with slope equal to $$-\frac{E_a}{R}$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 59
The d-electronic configuration of $$CoCl_4^{2-}$$ in tetrahedral crystal field is $$e^m t_2^n$$. The sum of m and number of unpaired electrons is
789
456
123
0.-
Clear All
NTA JEE Mains 24th january 2023 Shift 1 - Chemistry - Question 60
Number of moles of AgCl formed in the following reaction is _____.
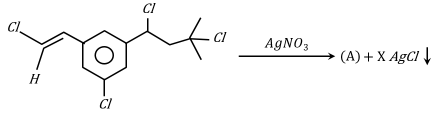
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




