NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 31
The number of radial node/s for 3p orbital is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 32
Given below are two statements:
Statement (I): Both metal and non-metal exist in p and d-block elements.
Statement (II): Non-metals have higher ionisation enthalpy and higher electronegativity than the metals.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 33
Given below are two statements:
Statement (I): A $$\pi$$ bonding MO has lower electron density above and below the inter-nuclear axis.
Statement (II): The $$\pi^*$$ antibonding MO has a node between the nuclei.
In light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 34
Select the compound from the following that will show intramolecular hydrogen bonding.
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 35
Solubility of calcium phosphate (molecular mass, M) in water is W g per 100 mL at 25°C. Its solubility product at 25°C will be approximately.
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 36
Match List - I with List - II.

Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 37
Given below are two statements:
Statement (I): $$SiO_2$$ and $$GeO_2$$ are acidic while SnO and PbO are amphoteric in nature.
Statement (II): Allotropic forms of carbon are due to property of catenation and $$p\pi - d\pi$$ bond formation.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 38
Which among the following has highest boiling point?
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 39
The set of meta directing functional groups from the following sets is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 40
The functional group that shows negative resonance effect is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 41
Lassaigne's test is used for detection of:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 42
In the given reactions identify A and B.
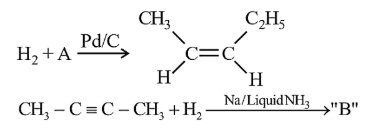
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 43
The strongest reducing agent among the following is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 44
The transition metal having highest 3rd ionisation enthalpy is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 45
Which of the following compounds show colour due to d-d transition?
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 46
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: In aqueous solutions $$Cr^{2+}$$ is reducing while $$Mn^{3+}$$ is oxidising in nature.
Reason R: Extra stability to half filled electronic configuration is observed than incompletely filled electronic configuration.
In the light of the above statement, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 47
Given below are two statements:
Statement (I): Dimethyl glyoxime forms a six membered covalent chelate when treated with $$NiCl_2$$ solution in presence of $$NH_4OH$$.
Statement (II): Prussian blue precipitate contains iron both in +2 and +3 oxidation states.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 48
$$[Co(NH_3)_6]^{3+}$$ and $$[CoF_6]^{3-}$$ are respectively known as:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 49
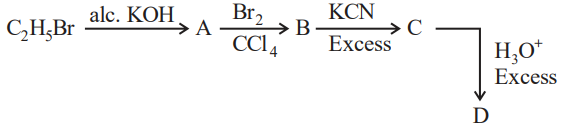
Acid D formed in above reaction is:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 50
Match List - I with List - II.

Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 51
10 mL of gaseous hydrocarbon on combustion gives 40 mL of $$CO_2(g)$$ and 50 mL of water vapour. Total number of carbon and hydrogen atoms in the hydrocarbon is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 52
For a certain reaction at 300 K, K = 10, then $$\Delta G°$$ for the same reaction is - ______ $$\times$$ $$10^{-1}$$ $$kJ mol^{-1}$$. (Given $$R = 8.314 \text{ J K}^{-1} \text{mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 53
Following Kjeldahl's method, 1 g of organic compound released ammonia, that neutralised 10 mL of $$2M$$ $$H_2SO_4$$. The percentage of nitrogen in the compound is ______ %.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 54
Total number of isomeric compounds (including stereoisomers) formed by monochlorination of 2-methylbutane is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 55
Mass of ethylene glycol (antifreeze) to be added to 18.6 kg of water to protect the freezing point at $$-24°C$$ is ______ kg (Molar mass in g mol$$^{-1}$$ for ethylene glycol 62, $$K_f$$ of water = $$1.86 \text{ K kg mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 56
The amount of electricity in Coulomb required for the oxidation of 1 mol of $$H_2O$$ to $$O_2$$ is ______ $$\times 10^5$$ C.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 57
Consider the following redox reaction: $$MnO_4^- + H^+ + H_2C_2O_4 \rightleftharpoons Mn^{2+} + H_2O + CO_2$$
The standard reduction potentials are given as below:
$$E^0_{MnO_4^-/Mn^{2+}} = +1.51$$ V; $$E^0_{CO_2/H_2C_2O_4} = -0.49$$ V
If the equilibrium constant of the above reaction is given as $$K_{eq} = 10^x$$, then the value of $$x$$ = ______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 58
The following data were obtained during the first order thermal decomposition of a gas A at constant volume:
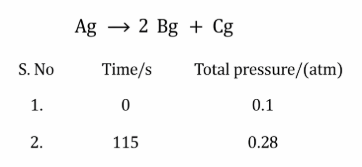
The rate constant of the reaction is ______ $$\times 10^{-2} \text{ s}^{-1}$$ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 59
Number of compounds which give reaction with Hinsberg's reagent is ______.
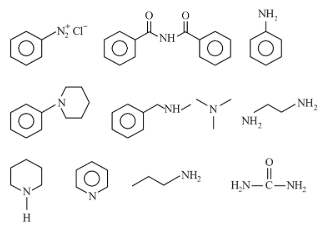
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2024 Shift 2 - Chemistry - Question 60
The number of tripeptides formed by three different amino acids using each amino acid once is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




