Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
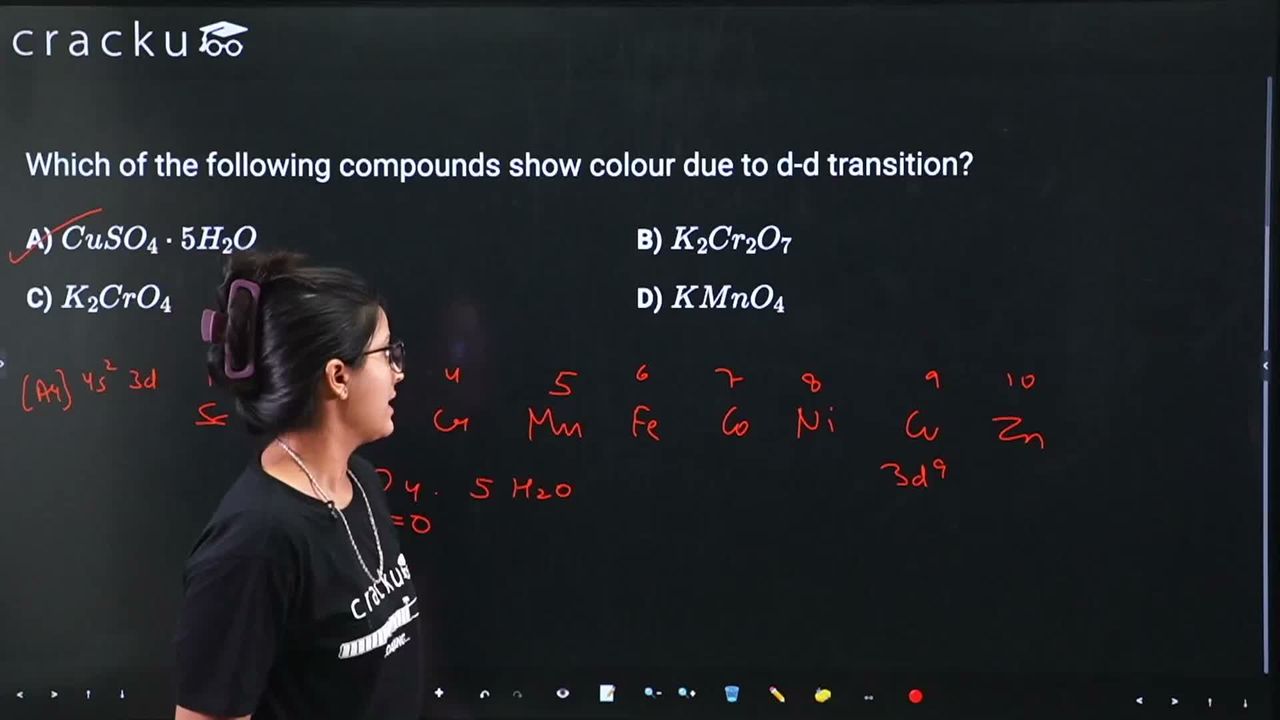
We need to identify which compound shows colour due to d-d transition among: $$CuSO_4 \cdot 5H_2O$$, $$K_2Cr_2O_7$$, $$K_2CrO_4$$, and $$KMnO_4$$.
d-d transitions involve the excitation of an electron from one d-orbital to another within the same metal ion. These transitions are typically weak (Laporte forbidden) and give rise to pale colours.
Charge transfer (CT) transitions involve transfer of electrons between ligand and metal orbitals. These are much more intense and give rise to deep, vivid colours.
$$CuSO_4 \cdot 5H_2O$$: Contains $$Cu^{2+}$$ with electronic configuration $$[Ar]3d^9$$. The $$Cu^{2+}$$ ion has an incomplete d-shell with one unpaired electron. In the hydrated form, $$[Cu(H_2O)_4]^{2+}$$ (approximately square planar/distorted octahedral), d-d transitions occur, giving the characteristic blue colour. This is a d-d transition.
$$K_2Cr_2O_7$$: Contains $$Cr^{6+}$$ ($$Cr_2O_7^{2-}$$) with configuration $$[Ar]3d^0$$. Since there are no d-electrons, d-d transitions are impossible. The orange colour is due to ligand-to-metal charge transfer (LMCT) from oxygen to chromium.
$$K_2CrO_4$$: Contains $$Cr^{6+}$$ ($$CrO_4^{2-}$$) with $$3d^0$$. Again, no d-electrons means no d-d transitions. The yellow colour is due to LMCT transition.
$$KMnO_4$$: Contains $$Mn^{7+}$$ ($$MnO_4^-$$) with $$3d^0$$. No d-electrons, so no d-d transitions. The intense purple colour is due to LMCT transition from oxygen to manganese.
Only $$CuSO_4 \cdot 5H_2O$$ shows colour due to d-d transition, as $$Cu^{2+}$$ has a $$3d^9$$ configuration allowing d-d electronic transitions.
The correct answer is $$CuSO_4 \cdot 5H_2O$$ (Option A).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
