NTA JEE Main 31st August 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 31
The incorrect expression among the following is:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 32
In which one of the following sets all species show disproportionation reaction?
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 33
Which one of the following statements is incorrect?
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 34
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Lithium salts are hydrated.
Reason (R): Lithium has higher polarising power than other alkali metal group members.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 35
Match List - I with List - II:
List - I (Metal ion) List - II (Group in Qualitative analysis)
(a) Mn$$^{2+}$$ (i) Group - III
(b) As$$^{3+}$$ (ii) Group - IIA
(c) Cu$$^{2+}$$ (iii) Group - IV
(d) Al$$^{3+}$$ (iv) Group - IIB
Choose the most appropriate answer from the options given below:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 36
Arrange the following conformational isomers of n-butane in order of their increasing potential energy:
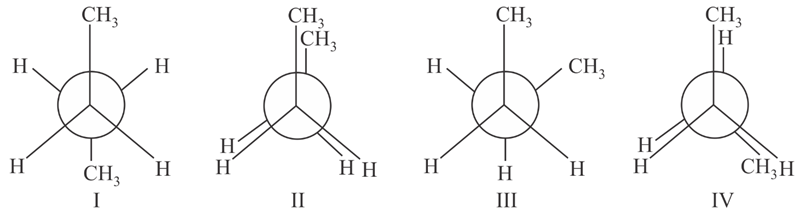
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 37
Identify correct A, B and C in the reaction sequence given below:
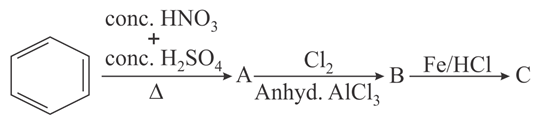
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 38
For the following sequence of reactions, the correct products are:
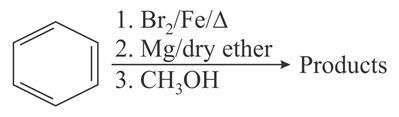
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 39
The deposition of X and Y on ground surfaces is referred as wet and dry depositions, respectively. X and Y are:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 40
Match List - I with List - II:
List - I (Parameter) List - II (Unit)
(a) Cell constant (i) S cm$$^2$$ mol$$^{-1}$$
(b) Molar conductivity (ii) Dimensionless
(c) Conductivity (iii) m$$^{-1}$$
(d) Degree of dissociation of electrolyte (iv) $$\Omega^{-1}$$ m$$^{-1}$$
Choose the most appropriate answer:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 41
The number of S = O bonds present in sulphurous acid, peroxodisulphuric acid and pyrosulphuric acid, respectively are:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 42
Which one of the following correctly represents the order of stability of oxides, X$$_2$$O; (X = halogen)?
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 43
The Eu$$^{2+}$$ ion is a strong reducing agent in spite of its ground state electronic configuration (outermost):
[Atomic number of Eu = 63]
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 44
Spin only magnetic moment in BM of FeCO$$_4$$C$$_2$$O$$_4^+$$ is:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 45
The major product of the following reaction is:
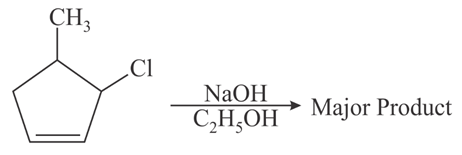
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 46
For the reaction given below:
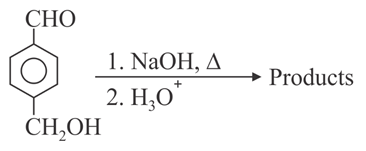
The compound which is not formed as a product in the reaction is a:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 47
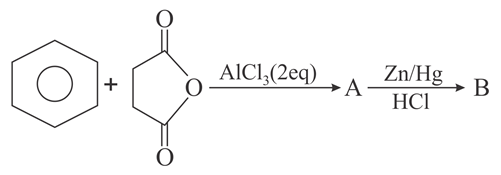
The structures of A and B formed in the following reaction are: Ph = -C$$_6$$H$$_5$$
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 48
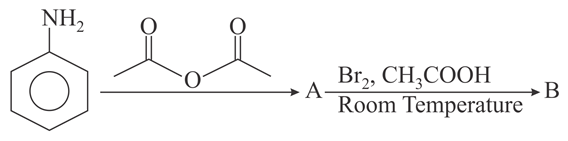
The major products A and B formed in the following reaction sequence are:
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 49
Which among the following is not a polyester?
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 50
Which of the following is NOT an example of fibrous protein?
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 51
The value of magnetic quantum number of the outermost electron of Zn$$^+$$ ion is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 52
According to molecular orbital theory, the number of unpaired electron(s) in O$$_2^{2-}$$ is _________.
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 53
The pH of a solution obtained by mixing 50 mL of 1 M HCl and 30 mL of 1 M NaOH is $$x \times 10^{-4}$$. The value of x is _________. (Nearest integer)
log 2.5 = 0.3979
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 54
The transformation occurring in Duma's method is given below
$$C_2H_7N + 2x + \frac{y}{2}CuO \rightarrow xCO_2 + \frac{y}{2}H_2O + \frac{z}{2}N_2 + 2x + \frac{y}{2}Cu$$
The value of y is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 55
The empirical formula for a compound with a cubic close packed arrangement of anions and with cations occupying all the octahedral sites in A$$_x$$B. The value of x is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 56
Sodium oxide reacts with water to produce sodium hydroxide. 20.0 g of sodium oxide is dissolved in 500 mL of water. Neglecting the change in volume, the concentration of the resulting NaOH solution is _________ $$\times 10^{-1}$$ M. (Nearest integer)
[Atomic mass : Na = 23.0, O = 16.0, H = 1.0]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 57
1.22 g of an organic acid is separately dissolved in 100 g of benzene (K$$_b$$ = 2.6 K kg mol$$^{-1}$$) and 100 g of acetone (K$$_b$$ = 1.7 K kg mol$$^{-1}$$). The acid is known to dimerize in benzene but remain as a monomer in acetone. The boiling point of the solution in acetone increases by 0.17°C. The increase in boiling point of solution in benzene in °C is $$x \times 10^{-2}$$. The value of x is _________. (Nearest integer)
[Atomic mass : C = 12.0, H = 1.0, O = 16.0]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 58
For the reaction A $$\rightarrow$$ B, the rate constant k (in s$$^{-1}$$) is given by
$$\log_{10} k = 20.35 - \frac{2.47 \times 10^3}{T}$$
The energy of activation in kJ mol$$^{-1}$$ is _________. (Nearest integer)
[Given : R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 59
CH$$_4$$ is adsorbed on 1 g charcoal at 0°C following the Freundlich adsorption isotherm. 10.0 mL of CH$$_4$$ is adsorbed at 100 mm of Hg, whereas 15.0 mL is adsorbed at 200 mm of Hg. The volume of CH$$_4$$ adsorbed at 300 mm of Hg is $$10^x$$ mL. The value of x is _________ $$\times 10^{-2}$$. (Nearest integer)
[Use $$\log_{10} 2 = 0.3010$$, $$\log_{10} 3 = 0.4771$$]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 2 - Chemistry - Question 60
In the electrolytic refining of blister copper, the total number of main impurities, from the following, removed as anode mud is _________.
Pb, Sb, Se, Te, Ru, Ag, Au and Pt
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)



