NTA JEE Main 24th June 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 31
If a rocket runs on a fuel ($$C_{15}H_{30}$$) and liquid oxygen, the weight of oxygen required and $$CO_2$$ released for every litre of fuel respectively are :
(Given : density of the fuel is $$0.756$$ g/mL)
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 32
Consider the following pairs of electrons
(A) (a) $$n = 3, l = 1, m_l = 1, m_s = +\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = 1, m_s = +\frac{1}{2}$$
(B) (a) $$n = 3, l = 2, m_l = -2, m_s = -\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = -1, m_s = -\frac{1}{2}$$
(C) (a) $$n = 4, l = 2, m_l = 2, m_s = +\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = 2, m_s = +\frac{1}{2}$$
The pairs of electrons present in degenerate orbitals is/are
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 33
For a reaction at equilibrium
$$A(g) \rightleftharpoons B(g) + \frac{1}{2}C(g)$$
the relation between dissociation constant ($$K$$), degree of dissociation ($$\alpha$$) and equilibrium pressure ($$p$$) is given by :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 34
The highest industrial consumption of molecular hydrogen is to produce compound of element :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 35
Which of the following statements are correct?
(A) Both LiCl and MgCl$$_2$$ are soluble in ethanol.
(B) The oxides Li$$_2$$O and MgO combine with excess of oxygen to give superoxide.
(C) LiF is less soluble in water than other alkali metal fluorides.
(D) Li$$_2$$O is more soluble in water than other alkali metal oxides.
Choose the most appropriate answer from the options given below
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 36
Identify the correct statement for $$B_2H_6$$ from those given below.
(A) In $$B_2H_6$$, all B $$-$$ H bonds are equivalent.
(B) In $$B_2H_6$$, there are four 3-centre-2-electron bonds.
(C) $$B_2H_6$$ is a Lewis acid.
(D) $$B_2H_6$$ can be synthesized from both $$BF_3$$ and $$NaBH_4$$.
(E) $$B_2H_6$$ is a planar molecule.
Choose the most appropriate answer from the options given below :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 37
Which of the following is an example of conjugated diketone?
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 38
In the given reaction sequence, the major product 'C' is :
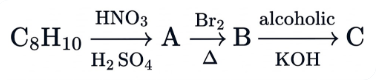
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 39
Given below are two statements:
Statement I : Emulsions of oil in water are unstable and sometimes they separate into two layers on standing.
Statement II : For stabilisation of an emulsion, excess of electrolyte is added.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 40
Match List - I with List - II :
| List-I | List-II | ||
|---|---|---|---|
| (A) | Sphalerite | (I) | FeCO$$_3$$ |
| (B) | Calamine | (II) | PbS |
| (C) | Galena | (III) | ZnCO$$_3$$ |
| (D) | Siderite | (IV) | ZnS |
Choose the most appropriate answer from the options given below :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 41
Given below are the oxides :
$$Na_2O, As_2O_3, N_2O, NO$$ and $$Cl_2O_7$$
Number of amphoteric oxides is :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 42
The most stable trihalide of nitrogen is :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 43
Which one of the following elemental forms is not present in the enamel of the teeth?
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 44
Match List - I with List - II :
| List-I | List-II | ||
|---|---|---|---|
| (A) | $$[PtCl_4]^{2-}$$ | (I) | $$sp^3d$$ |
| (B) | $$BrF_5$$ | (II) | $$d^2sp^3$$ |
| (C) | $$PCl_5$$ | (III) | $$dsp^2$$ |
| (D) | $$[Co(NH_3)_6]^{3+}$$ | (IV) | $$sp^3d^2$$ |
Choose the most appropriate answer from the options given below
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 45
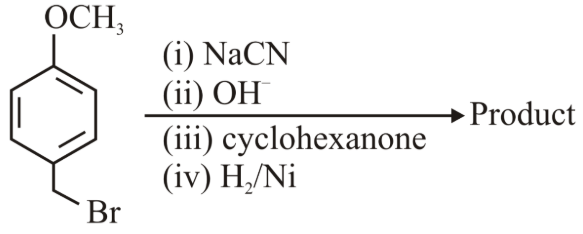
The major product of the above reactions is
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 46
Two statements are given below :
Statement I : The melting point of monocarboxylic acid with even number of carbon atoms is higher than that of with odd number of carbon atoms acid immediately below and above it in the series.
Statement II : The solubility of monocarboxylic acids in water decreases with increase in molar mass.
Choose the most appropriate option :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 47
Which of the following is an example of polyester?
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 48
Which of the following is not a broad spectrum antibiotic?
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 49
During the qualitative analysis of salt with cation $$y^{2+}$$, addition of a reagent (X) to alkaline solution of the salt gives a bright red precipitate. The reagent (X) and the cation ($$y^{2+}$$) present respectively are :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 50
A polysaccharide 'X' on boiling with dil $$H_2SO_4$$ at $$393$$ K under $$2 - 3$$ atm pressure yields 'Y' 'Y' on treatment with bromine water gives gluconic acid. 'X' contains $$\beta$$-glycosidic linkages only. Compound 'X' is :
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 51
$$2O_3(g) \rightleftharpoons 3O_2(g)$$
At $$300$$ K, ozone is fifty percent dissociated. The standard free energy change at this temperature and $$1$$ atm pressure is $$(-)$$ ______ J mol$$^{-1}$$. (Nearest integer)
[Given: $$\ln 1.35 = 0.3$$ and $$R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 52
A $$0.166$$ g sample of an organic compound was digested with conc. $$H_2SO_4$$ and then distilled with NaOH. The ammonia gas evolved was passed through $$50.0$$ mL of $$0.5$$ N $$H_2SO_4$$. The used acid required $$30.0$$ mL of $$0.25$$ N NaOH for complete neutralization. The mass percentage of nitrogen in the organic compound is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 53
Number of electrophilic centres in the given compound is ______
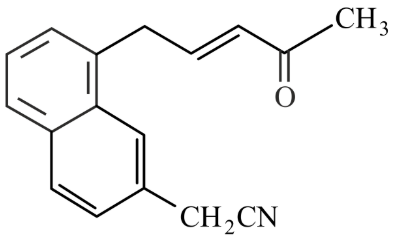
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 54
The major product 'A' of the following given reaction has $$sp^2$$ hybridized carbon atoms.
$$2,7\text{-Dimethyl-2,6-octadiene} \xrightarrow{H^+} A$$ (Major Product)
The number of $$sp^2$$ hybridized carbon atoms in 'A' is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 55
Atoms of element X form hcp lattice and those of element Y occupy $$\frac{2}{3}$$ of its tetrahedral voids. The percentage of element X in the lattice is (Nearest integer) ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 56
The osmotic pressure of blood is $$7.47$$ bar at $$300$$ K. To inject glucose to a patient intravenously, it has to be isotonic with blood. The concentration of glucose solution in gL$$^{-1}$$ is (Molar mass of glucose $$= 180$$ g mol$$^{-1}$$, $$R = 0.083$$ Lbar$$^{-1}$$ mol$$^{-1}$$) (Nearest integer) ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 57
The cell potential for the following cell $$Pt|H_2(g)|H^+(aq)||Cu^{2+}(0.01 \text{ M})|Cu(s)$$ is $$0.576$$ V at $$298$$ K. The pH of the solution is (Nearest integer) ______
(Given : $$E^{\circ}_{Cu^{2+}/Cu} = 0.34$$ V and $$\frac{2.303RT}{F} = 0.06$$ V)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 58
The rate constants for decomposition of acetaldehyde have been measured over the temperature range $$700 - 1000$$ K. The data has been analysed by plotting $$\ln k$$ vs $$\frac{10^3}{T}$$ graph. The value of activation energy for the reaction is ______ kJ mol$$^{-1}$$. (Nearest integer) (Given : $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$)
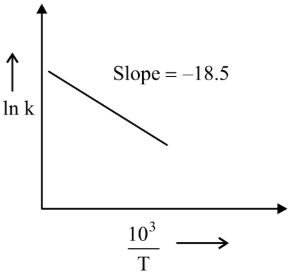
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 59
The difference in oxidation state of chromium in chromate and dichromate salts is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Chemistry - Question 60
In the cobalt-carbonyl complex : $$[Co_2(CO)_8]$$, number of Co $$-$$ Co bonds is "X" and terminal CO ligands is "Y". $$X + Y =$$ ______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




