Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
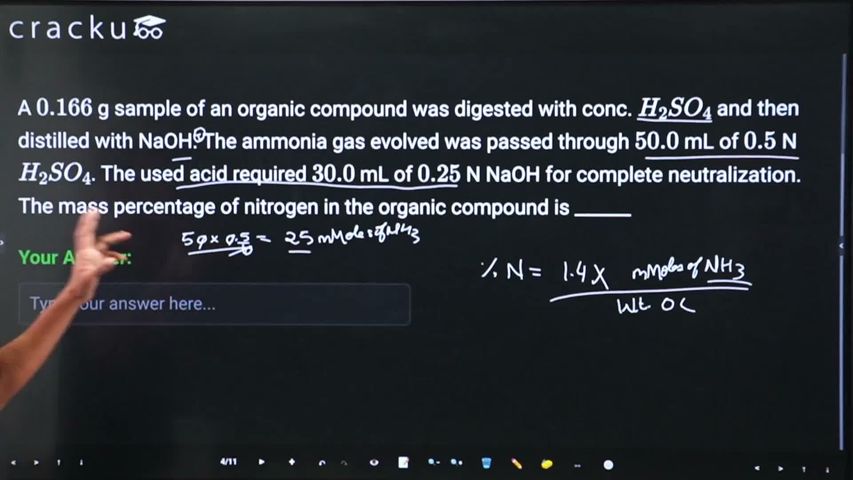
A $$0.166$$ g sample of an organic compound was digested with conc. $$H_2SO_4$$ and then distilled with NaOH. The ammonia gas evolved was passed through $$50.0$$ mL of $$0.5$$ N $$H_2SO_4$$. The used acid required $$30.0$$ mL of $$0.25$$ N NaOH for complete neutralization. The mass percentage of nitrogen in the organic compound is ______
Correct Answer: 63
In Kjeldahl's method, the mass percentage of nitrogen (%N) in an organic compound is given by the formula:
%N=Mass of the organic compound (in g)1.4×Milliequivalents of acid consumed by NH3
Where:
Milliequivalents (meq)=Normality (N)×Volume (in mL)
If we consider the 7.5 meq to be the leftover unreacted acid, then the acid consumed by ammonia would be:
25.0 meq−7.5 meq=17.5 meq
Substituting this into the percentage formula yields:
%N=0.1661.4×17.5≈147.6%
Since a mass percentage greater than 100% is physically impossible, the terminology "the used acid required" indicates that the milliequivalents of acid effectively consumed by the ammonia gas corresponds directly to the titration value:
Milliequivalents of acid consumed by NH3=meqNaOH=7.5 meq
Substituting the values into the Kjeldahl's formula:
%N=0.1661.4×7.5%N=0.16610.5≈63.25%
Rounding to the nearest integer, we get:
63%
The mass percentage of nitrogen in the organic compound is 63% (or 63.25%).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
