Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
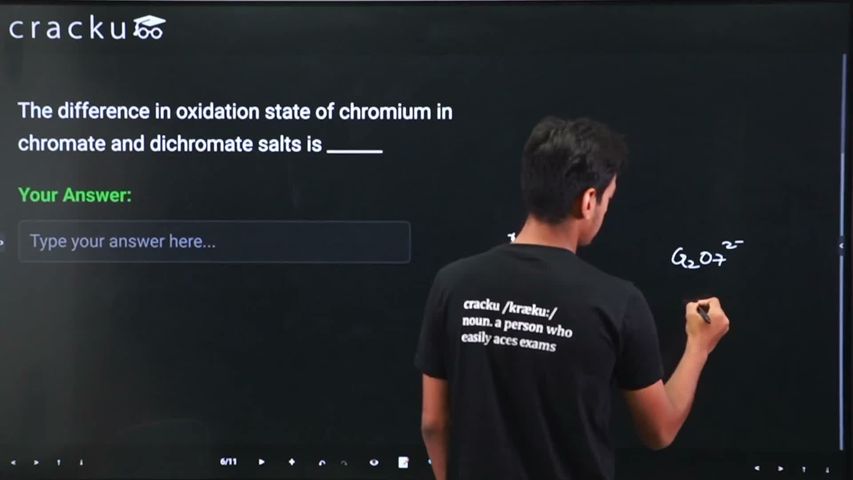
The difference in oxidation state of chromium in chromate and dichromate salts is ______
Correct Answer: 0
We need to find the difference in oxidation state of chromium in chromate (CrO$$_4^{2-}$$) and dichromate (Cr$$_2$$O$$_7^{2-}$$) salts. Let the oxidation state of Cr in chromate be $$x$$.
$$ x + 4(-2) = -2 $$
$$ x - 8 = -2 $$
$$ x = +6 $$
Let the oxidation state of Cr in dichromate be $$y$$.
$$ 2y + 7(-2) = -2 $$
$$ 2y - 14 = -2 $$
$$ 2y = 12 $$
$$ y = +6 $$
The difference in oxidation states:
$$ x - y = +6 - (+6) = 0 $$
Therefore, the difference in oxidation state of chromium in chromate and dichromate salts is 0.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
