NTA JEE Main 10th January 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 31
The 71$$^{st}$$ electron of an element X with an atomic number of 71 enters the orbital:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 32
The ground state energy of a hydrogen atom is $$-13.6$$ eV. The energy of second excited state of He$$^+$$ ion in eV is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 33
The process with negative entropy change is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 34
An ideal gas undergoes isothermal compression from 5 m$$^3$$ to 1 m$$^3$$ against a constant external pressure of 4 N m$$^{-2}$$. The heat released in this process is 24 J mol$$^{-1}$$ K$$^{-1}$$ and is used to increase the pressure of 1 mole of Al. The temperature of Al increases by:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 35
5.1 g $$NH_4SH$$ is introduced in 3.0 L evacuated flask at 327$$^{\circ}$$C. 30% of the solid $$NH_4SH$$ is decomposed to $$NH_3$$ and $$H_2S$$ as gases. The $$K_P$$ of the reaction at 327$$^{\circ}$$C is: ($$R = 0.082$$ L atm mol$$^{-1}$$ K$$^{-1}$$, Molar mass of S = 32 g mol$$^{-1}$$, Molar mass of N = 14 g mol$$^{-1}$$)
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 36
In the reaction of oxalate with permanganate in acidic medium, the number of electrons involved in producing one molecule of CO$$_2$$ is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 37
The number of 2-centre-2-electron and 3-centre-2-electron bonds in $$B_2H_6$$, respectively, are:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 38
What is the IUPAC name of the following compound?
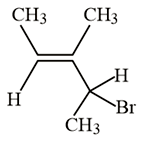
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 39
What will be the major product in the following mononitration reaction?
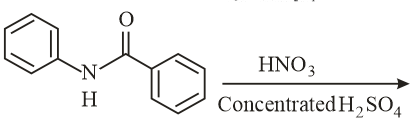
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 40
The reaction that is not involved in the ozone layer depletion mechanism in the stratosphere is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 41
A compound of formula $$A_2B_3$$ has the HCP lattice. Which atom forms the HCP lattice and what fraction of the tetrahedral voids are occupied by the other atoms?
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 42
The amount of sugar ($$C_{12}H_{22}O_{11}$$) required to prepare 2L of its 0.1 M aqueous solution is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 43
The elevation in boiling point for 1 molal solution of glucose is 2 K. The depression in freezing point for 2 molal solution of glucose in the same solvent is 2 K. The relation between $$K_b$$ and $$K_f$$ is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 44
In the cell, Pt(s)|H$$_2$$(g, 1 bar)|HCl(aq)|AgCl(s)|Ag(s)|Pt(s), the cell potential is 0.92 V when a $$10^{-6}$$ molar HCl solution is used. The standard electrode potential of Ag|AgCl|Cl$$^-$$ electrode is: (Given, $$\frac{2.303RT}{F} = 0.06$$ V at 298 K)
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 45
For an elementary chemical reaction, $$A_2 \underset{k_{-1}}{\overset{k_1}{\rightleftharpoons}} 2A$$, the expression for $$\frac{d[A]}{dt}$$ is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 46
The haemoglobin and the gold sol are examples of:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 47
The electrolytes usually used in the electroplating of gold and silver, respectively, are:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 48
Among the following reactions of hydrogen with halogens, the one that requires a catalyst is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 49
The pair that contains two $$P - H$$ bonds in each of the oxoacids is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 50
Sodium metal on dissolution in liquid ammonia gives a deep blue solution due to the formation of:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 51
A reaction of cobalt (III) chloride and ethylenediamine in a 1 : 2 mole ratio generates two isomeric products A (violet-coloured) and B (green-coloured). A can show optical activity, but, B is optically inactive. What type of isomers do A and B represent?
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 52
The difference in the number of unpaired electrons of a metal ion in its high-spin and low-spin octahedral complexes is two. The metal ion is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 53
The major product of the following reaction is:
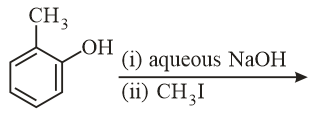
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 54
The major product obtained in the following reaction is:
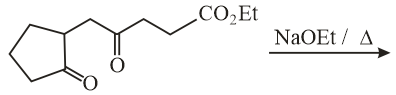
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 55
The major product of the following reaction is:
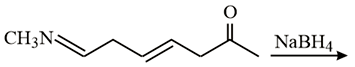
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 56
Which is the most suitable reagent for the following transformation?
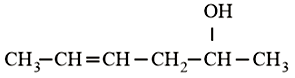
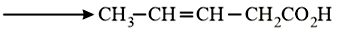
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 57
An aromatic compound 'A' having molecular formula $$C_7H_6O_2$$, on treating with aqueous ammonia and heating forms compound 'B'. The compound 'B' on reaction with molecular bromine and potassium hydroxide provides compound 'C' having molecular formula $$C_6H_7N$$. The structure of 'A' is:
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 58
The major product of the following reaction is:
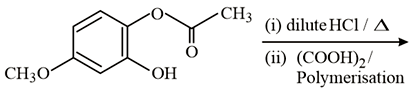
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 59
Which of the following tests cannot be used for identifying amino acids?
NTA JEE Main 10th January 2019 Shift 2 - Chemistry - Question 60
The correct match between item I and item II is:
Item I (Compound) Item II (Reagent)
a. Lysine p. 1-naphthol
b. Furfural q. Ninhydrin
c. Benzyl alcohol r. KMnO$$_4$$
d. Styrene s. Ceric ammonium nitrate
.webp)
.webp)
.webp)
.webp)




