Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A sample of gas expands from $$V_1$$ to $$V_2$$. In which of the following, the work done will be greatest?
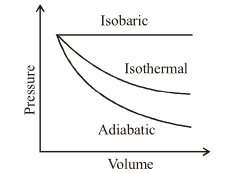
The work done is greatest for the isobaric process because it corresponds to the largest area under the P-V curve during expansion. Since the pressure remains constant and highest throughout this process, it encloses more area than the decreasing curves of the isothermal and adiabatic processes.
Create a FREE account and get:
Educational materials for JEE preparation
