Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
On reaction with stronger oxidizing agent like $$KIO_4$$, hydrogen peroxide oxidizes with the evolution of $$O_2$$. The oxidation number of I in $$KIO_4$$ changes to
Correct Answer: 5
We are told that $$KIO_4$$ (potassium periodate) is a stronger oxidizing agent that reacts with hydrogen peroxide ($$H_2O_2$$), causing $$H_2O_2$$ to get oxidized with the evolution of $$O_2$$. We need to find the oxidation number of iodine in the product after this reaction.
In $$KIO_4$$, potassium has an oxidation state of +1 and each oxygen has -2. So the oxidation state of iodine is: $$+1 + x + 4(-2) = 0$$, which gives $$x = +7$$. So iodine starts with an oxidation state of +7.
The reaction is:
$$KIO_4 + H_2O_2 \rightarrow KIO_3 + H_2O + O_2$$
Here, $$H_2O_2$$ is oxidized (oxygen goes from -1 in $$H_2O_2$$ to 0 in $$O_2$$), while $$KIO_4$$ is reduced. The iodine in $$KIO_4$$ (oxidation state +7) is reduced to iodine in $$KIO_3$$ (potassium iodate).
In $$KIO_3$$: $$+1 + x + 3(-2) = 0$$, which gives $$x = +5$$.
So the oxidation number of iodine changes from +7 in $$KIO_4$$ to +5 in $$KIO_3$$.
Hence, the correct answer is 5.
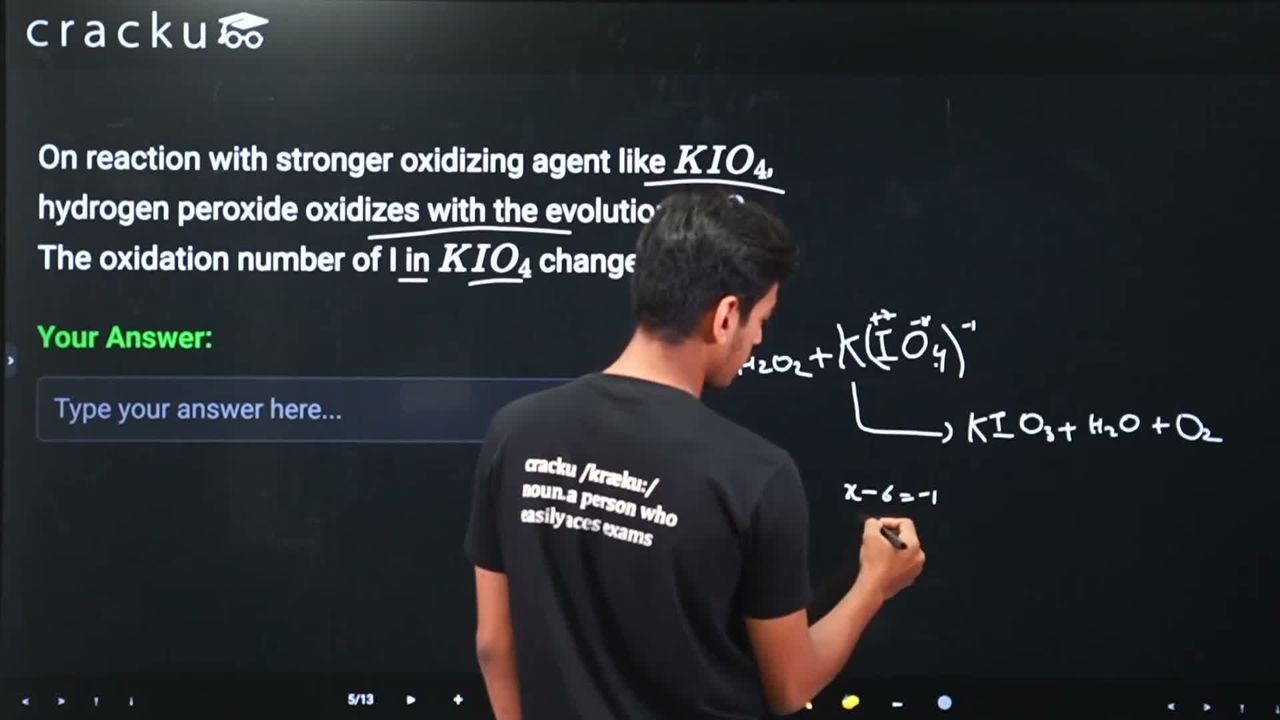
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
