Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
At 600 K, 2 mol of NO are mixed with 1 mol of $$O_2$$.
$$2NO(g) + O_2(g) \rightleftharpoons 2NO_2(g)$$
The reaction occurring as comes to equilibrium under a total pressure of 1 atm. Analysis of the system shows that 0.6 mol of oxygen are present at equilibrium. The equilibrium constant for the reaction is _____
Correct Answer: 2
We are given the reaction $$2NO(g) + O_2(g) \rightleftharpoons 2NO_2(g)$$ at 600 K. Initially, 2 mol of NO and 1 mol of $$O_2$$ are mixed, and the total pressure at equilibrium is 1 atm. At equilibrium, 0.6 mol of $$O_2$$ is present. We need to find the equilibrium constant $$K_p$$.
Let us set up an ICE table. Let $$x$$ be the moles of $$O_2$$ consumed.
Since 1 mol of $$O_2$$ was initially present and 0.6 mol remains at equilibrium, $$x = 1 - 0.6 = 0.4$$ mol of $$O_2$$ has been consumed.
From the stoichiometry, 2 mol of NO are consumed for every 1 mol of $$O_2$$, so moles of NO consumed = $$2 \times 0.4 = 0.8$$ mol. Also, 2 mol of $$NO_2$$ are formed for every 1 mol of $$O_2$$ consumed, so moles of $$NO_2$$ formed = $$2 \times 0.4 = 0.8$$ mol.
At equilibrium:
$$n_{NO} = 2 - 0.8 = 1.2 \text{ mol}$$
$$n_{O_2} = 1 - 0.4 = 0.6 \text{ mol}$$
$$n_{NO_2} = 0 + 0.8 = 0.8 \text{ mol}$$
Total moles at equilibrium: $$n_{total} = 1.2 + 0.6 + 0.8 = 2.6 \text{ mol}$$
Now we calculate the mole fractions and partial pressures (using $$P_{total} = 1$$ atm):
$$P_{NO} = \frac{1.2}{2.6} \times 1 = \frac{1.2}{2.6} \text{ atm}$$
$$P_{O_2} = \frac{0.6}{2.6} \times 1 = \frac{0.6}{2.6} \text{ atm}$$
$$P_{NO_2} = \frac{0.8}{2.6} \times 1 = \frac{0.8}{2.6} \text{ atm}$$
The equilibrium constant expression is:
$$K_p = \frac{(P_{NO_2})^2}{(P_{NO})^2 \cdot P_{O_2}}$$
Substituting:
$$K_p = \frac{\left(\frac{0.8}{2.6}\right)^2}{\left(\frac{1.2}{2.6}\right)^2 \times \frac{0.6}{2.6}}$$
$$= \frac{\frac{0.64}{6.76}}{\frac{1.44}{6.76} \times \frac{0.6}{2.6}}$$
$$= \frac{0.64}{1.44 \times \frac{0.6}{2.6}}$$
$$= \frac{0.64 \times 2.6}{1.44 \times 0.6}$$
$$= \frac{1.664}{0.864}$$
$$\approx 1.926$$
Rounding to the nearest integer, $$K_p \approx 2$$.
Hence, the correct answer is 2.
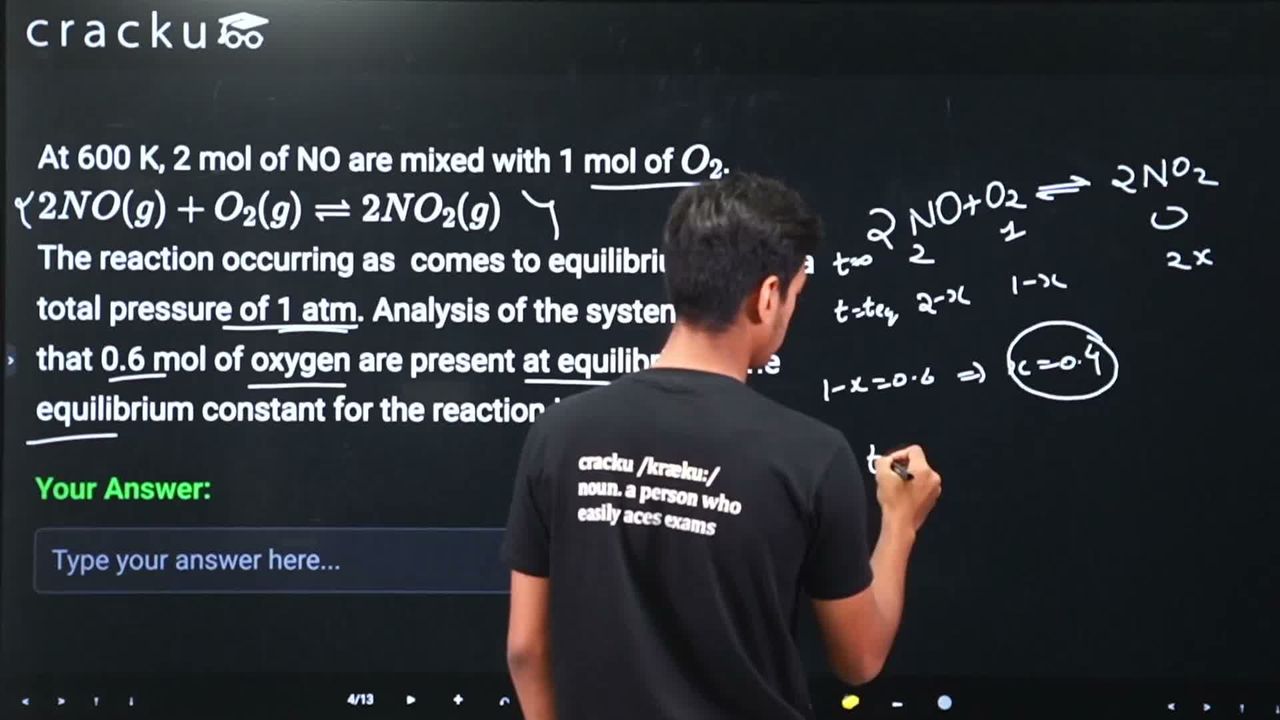
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
