Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The major products obtained from the reactions in List-II are the reactants for the named reactions mentioned in List-I. Match List-I with List-II and choose the correct option.
| List-I | List-II | ||
|---|---|---|---|
| (P) | Etard reaction | (1) | Acetophenone $$\xrightarrow{Zn-Hg, HCl}$$ |
| (Q) | Gattermann reaction | (2) | Toluene $$\xrightarrow[\text{(ii) } SOCl_2]{\text{(i) } KMnO_4, KOH, \Delta}$$ |
| (R) | Gattermann-Koch reaction | (3) | 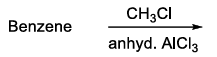 |
| (S) | Rosenmund reduction | (4) | 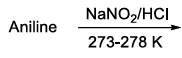 |
| (5) | Phenol $$\xrightarrow{Zn, \Delta}$$ | ||
The problem gives four named organic reactions (List-I) and five synthetic sequences whose final products will act as starting materials for those named reactions (List-II).
To find the correct pairing, we must first determine the major product of every sequence (1) - (5) and then recall which starting material each named reaction requires.
Step 1 : Identify the products of List-II
(1) $$\text{Acetophenone }\big(C_6H_5COCH_3\big) \xrightarrow[\text{conc. }HCl]{Zn\text{-}Hg}$$
The reagent pair $$Zn\text{-}Hg/HCl$$ performs Clemmensen reduction, converting the carbonyl group $$C=O$$ to $$CH_2$$.
Product : $$C_6H_5CH_2CH_3$$ (ethylbenzene).
(2) $$\text{Toluene }\big(C_6H_5CH_3\big)\xrightarrow{\text{KMnO}_4/KOH,\,\Delta}\ C_6H_5COOK\xrightarrow{H^+}\ C_6H_5COOH\xrightarrow{SOCl_2}\ C_6H_5COCl$$ Potassium permanganate oxidises the side-chain methyl to a carboxylate; acidification gives benzoic acid, which $$SOCl_2$$ changes to benzoyl chloride $$C_6H_5COCl$$.
(3) $$\text{Benzene }\xrightarrow{CH_3Cl,\ \text{anhyd. }AlCl_3}$$
This is Friedel-Crafts alkylation, introducing a methyl group.
Product : $$C_6H_5CH_3$$ (toluene).
(4) $$\text{Aniline }\big(C_6H_5NH_2\big)\xrightarrow{NaNO_2/HCl,\ 273-278\,\text{K}}$$ Diazotisation forms benzenediazonium chloride $$C_6H_5N_2^+Cl^-$$.
(5) $$\text{Phenol }\xrightarrow{Zn,\ \Delta}$$ Distillation of phenol with zinc removes oxygen, giving benzene $$C_6H_6$$.
Step 2 : Recall the required reactant for each named reaction in List-I
P Etard reaction : oxidises the benzylic $$-CH_3$$ group of toluene to an aldehyde. Required reactant → toluene.
Q Gattermann reaction : converts an aromatic diazonium salt to the corresponding chloro- or bromo-benzene using $$CuCl/CuBr$$. Required reactant → benzenediazonium chloride.
R Gattermann-Koch reaction : formylates benzene to benzaldehyde using $$CO/HCl$$ with $$AlCl_3/CuCl$$. Required reactant → benzene.
S Rosenmund reduction : hydrogenolyses an acyl chloride to an aldehyde using $$H_2/Pd\,\text{(poisoned)}$$. Required reactant → benzoyl (acyl) chloride.
Step 3 : Match List-I with List-II
P (Etard) needs toluene → obtained in (3).
Q (Gattermann) needs benzenediazonium chloride → obtained in (4).
R (Gattermann-Koch) needs benzene → obtained in (5).
S (Rosenmund) needs benzoyl chloride → obtained in (2).
Thus the correct matching is:
P → 3, Q → 4, R → 5, S → 2.
The option containing this sequence is Option D: P → 3; Q → 4; R → 5; S → 2.
Create a FREE account and get:
Educational materials for JEE preparation
