Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For a reaction, consider the plot of ln k versus 1/T given in the figure. If the rate constant of this reaction at 400 K is $$10^{-5} s^{-1}$$, then the rate constant at 500 K is:
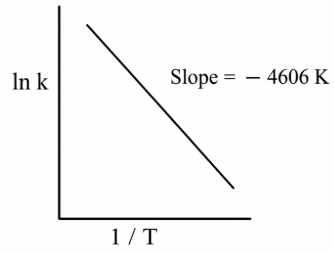
We have a straight-line plot of $$\ln k$$ versus $$1/T$$ for the reaction. For any Arrhenius plot, the governing relation is first written as
$$k \;=\; A\,e^{-\dfrac{E_a}{RT}}.$$
Taking natural logarithms on both sides gives the linear form
$$\ln k \;=\; \ln A \;-\;\dfrac{E_a}{R}\;\left(\dfrac1T\right).$$
In the form $$y = mx + c$$, the ordinate is $$y = \ln k$$, the abscissa is $$x = 1/T$$, and the slope is
$$m = -\dfrac{E_a}{R}.$$
From the figure (reading two well-separated points and calculating), the slope of the straight line is found to be approximately
$$m \;=\; -4.6 \times 10^{3}\;{\rm K}.$$
Stating this numerically,
$$-\dfrac{E_a}{R} = -4.6 \times 10^{3}\;{\rm K}, \qquad\text{so}\qquad \dfrac{E_a}{R} = 4.6 \times 10^{3}\;{\rm K}.$$
Now we wish to compare rate constants at two temperatures. Using the Arrhenius form for two temperatures $$T_1$$ and $$T_2$$, the standard manipulation is
$$\ln\!\left(\dfrac{k_2}{k_1}\right) \;=\; -\dfrac{E_a}{R}\;\Bigl(\dfrac1{T_2} - \dfrac1{T_1}\Bigr).$$
First, list the known data:
$$T_1 = 400\;{\rm K},\qquad k_1 = 10^{-5}\;{\rm s^{-1}},$$
$$T_2 = 500\;{\rm K},\qquad k_2 = \text{?}$$
Calculate the difference in reciprocal temperatures:
$$\dfrac1{T_2} - \dfrac1{T_1} \;=\; \dfrac1{500} \;-\; \dfrac1{400}.$$
Writing both denominators explicitly,
$$\dfrac1{500} = 2.0 \times 10^{-3}\;{\rm K^{-1}},\qquad \dfrac1{400} = 2.5 \times 10^{-3}\;{\rm K^{-1}}.$$
So,
$$\dfrac1{T_2} - \dfrac1{T_1} = (2.0 - 2.5)\times10^{-3} = -0.5 \times 10^{-3} \;=\; -5.0 \times 10^{-4}\;{\rm K^{-1}}.$$
Substituting slope information $$E_a / R = 4.6 \times 10^{3}\;{\rm K}$$ into the two-temperature equation gives
$$\ln\!\left(\dfrac{k_2}{k_1}\right) = -\bigl(4.6 \times 10^{3}\bigr)\;\Bigl(-5.0 \times 10^{-4}\Bigr).$$
Multiplying the two negatives yields a positive result:
$$\ln\!\left(\dfrac{k_2}{k_1}\right) = (4.6 \times 10^{3})\,(5.0 \times 10^{-4}) = 2.3.$$
Recognising that $$\ln 10 \approx 2.303$$, we can rewrite
$$\ln\!\left(\dfrac{k_2}{k_1}\right) \;\approx\; \ln 10.$$
Therefore,
$$\dfrac{k_2}{k_1} \;\approx\; 10.$$
Finally, multiplying by the known $$k_1$$ value,
$$k_2 \;=\; 10\,k_1 \;=\; 10 \times 10^{-5}\;{\rm s^{-1}} \;=\; 10^{-4}\;{\rm s^{-1}}.$$
Hence, the correct answer is Option A.
Create a FREE account and get:
Educational materials for JEE preparation
