Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A cylinder containing an ideal gas (0.1 mol of 1.0 dm$$^3$$) is in thermal equilibrium with a large volume of 0.5 molal aqueous solution of ethylene glycol at its freezing point. If the stoppers $$S_1$$ and $$S_2$$ (as shown in the figure) are suddenly withdrawn, the volume of the gas in litres after equilibrium is achieved will be ___________.
(Given, $$K_f$$(water) $$= 2.0$$ K kg mol$$^{-1}$$, R $$= 0.08$$ dm$$^3$$ atm K$$^{-1}$$ mol$$^{-1}$$)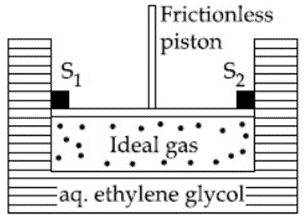
Correct Answer: 2.18
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.