Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Identify the major products A and B respectively in the following reactions of phenol
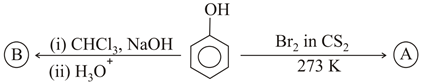
Phenol is an electron-rich aromatic compound because the lone pair on oxygen is in resonance with the ring. However, whenever phenol is first converted into a phenoxide ion (or any of its O-derivatives such as esters or ethers), the oxygen atom loses its ability to donate electron density as strongly as in phenol itself. Keeping this in mind, let us analyse the two consecutive reactions.
Case 1: Formation of $$A$$ (Acylation of phenol)
The first reagent given is an acid chloride / acetic anhydride in the presence of a base or pyridine (Schotten-Baumann / Friedel-Crafts acylation conditions for alcohols). The general reaction is:
$$Ar\!-\!OH \;+\; CH_3COCl \;\xrightarrow[\text{base}]{\,} Ar\!-\!OCOCH_3 \;+\; HCl$$
Thus phenol is converted into an ester, namely phenyl acetate (also written as $$O$$-acetyl phenol). Therefore, $$A$$ is
$$A = \text{Phenyl acetate : } C_6H_5OCOCH_3$$
Case 2: Conversion of $$A$$ to $$B$$ (Electrophilic nitration/bromination on the aromatic ring)
The second step involves treating compound $$A$$ with a strong electrophile (such as $$HNO_3/H_2SO_4$$ or $$Br_2/FeBr_3$$ as specified in the question). Because the oxygen is now tied up in an ester linkage, its +M (mesomeric) effect toward the ring is weaker than in free phenol, but it is still an ortho, para-directing group. Steric factors make the para product predominate.
If the electrophile is $$NO_2^{+}$$ (from conc. $$HNO_3/H_2SO_4$$), the reaction is:
$$C_6H_5OCOCH_3 \xrightarrow{\;HNO_3/H_2SO_4\;} NO_2\!-\!C_6H_4OCOCH_3 + H_2O$$
The major product will have the $$NO_2$$ group at the para position with respect to the ester functionality.
Hence,
$$B = \text{p-Nitrophenyl acetate : } NO_2\!-\!C_6H_4OCOCH_3$$
Summarising, $$A = \text{Phenyl acetate}$$ and $$B = \text{p-Nitrophenyl acetate}$$.
Among the given answer choices, this pair corresponds to Option D (Option 4). Therefore, the correct answer is Option D.
Educational materials for JEE preparation
