Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the given reaction:
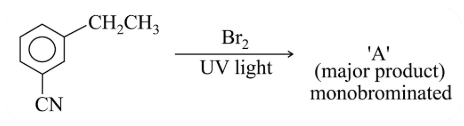
What is A?
The given sequence involves the two classic steps used to replace the -NH2 group of aniline by a halogen:
(i) Diazotisation of aniline.
(ii) Sandmeyer reaction of the diazonium salt with a cuprous halide.
Step 1 — Diazotisation
Aniline is treated at 0-5 °C with $$NaNO_2$$ and dil. $$HCl$$.
The nitrous acid generated in situ converts the -NH2 group into a diazonium group.
$$C_6H_5NH_2 + NaNO_2 + 2HCl \;\xrightarrow[0-5^{\circ}C]{}\; C_6H_5N_2^{+}Cl^{-} + NaCl + 2H_2O$$
The product after Step 1 is benzene diazonium chloride.
Step 2 — Sandmeyer reaction
When benzene diazonium chloride is warmed in the presence of cuprous bromide $$Cu_2Br_2/CuBr$$ (or simply written as $$CuBr$$) in hydrobromic acid, the diazonium group is replaced by bromine. Nitrogen gas is liberated.
$$C_6H_5N_2^{+}Cl^{-} + CuBr + HBr \;\xrightarrow{\,\Delta\,}\; C_6H_5Br + N_2 + CuCl + HCl$$
The organic product obtained is bromobenzene.
Therefore, compound A formed in the given reaction sequence is bromobenzene, which corresponds to Option B (Correct Answer: 2).
Educational materials for JEE preparation
