Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The major product of the following reaction is: (compound with Cl reacting with NaOEt/heat)
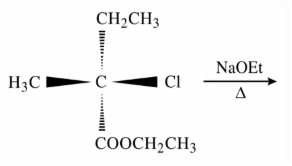
We have the substrate $$CH_3CHClCH_2CO_2CH_2CH_3$$ (ethyl 3-chlorobutanoate). The chlorine atom is attached to the carbon that is α to the ester carbonyl, while two different sets of β-hydrogen atoms are available for an E2 elimination.
Because the reagent is $$NaOEt$$ in ethanol and the reaction is carried out under heat, we immediately recognise that a strong base is present and that the reaction will proceed predominantly through the E2 mechanism rather than substitution. In an E2 process the base removes a β-hydrogen that is anti-periplanar to the leaving group, the C-H and C-Cl bonds break simultaneously and a C=C double bond is formed. The general E2 equation can be written as
$$\text{Base} + RCH_2CH_2Cl \;\longrightarrow\; RCH=CH_2 + Cl^- + \text{Base-H}$$
In our molecule we label the carbon bearing chlorine as $$C_\alpha$$ and the two adjacent carbons as $$C_{\beta 1}$$ (the CH2 group next to the ester) and $$C_{\beta 2}$$ (the terminal CH3 group). Hence the two distinct elimination possibilities are:
1. Removal of a hydrogen from $$C_{\beta 1}$$ (the CH2 between the Cl-bearing carbon and the carbonyl).
2. Removal of a hydrogen from $$C_{\beta 2}$$ (the terminal CH3).
We now write both possible eliminations explicitly.
Path 1 : β-H from $$C_{\beta 1}$$
The base abstracts one of the two hydrogens on $$C_{\beta 1}$$:
$$\begin{aligned} CH_3CHClCH_2CO_2CH_2CH_3 &\xrightarrow[\text{E2}]{NaOEt/\,\Delta} CH_3CH = CHCO_2CH_2CH_3 + Cl^- + HOEt \end{aligned}$$
The double bond produced is between $$C_\alpha$$ and $$C_{\beta 1}$$, giving us ethyl but-2-enoate (also called ethyl crotonate). This alkene is (i) trisubstituted and therefore more highly substituted, and (ii) conjugated with the carbonyl group of the ester, giving it extra resonance stabilisation. The resonance can be shown as
$$CH_3CH = CHCO_2Et \;\rightleftharpoons\; CH_3CH^- - CH = C^+O_2Et$$
Such conjugation lowers the overall energy of the molecule, favouring its formation.
Path 2 : β-H from $$C_{\beta 2}$$
If instead the base removes a hydrogen from the terminal CH3, we obtain
$$\begin{aligned} CH_3CHClCH_2CO_2CH_2CH_3 &\xrightarrow[\text{E2}]{NaOEt/\,\Delta} CH_3CH_2CH = CHCO_2CH_2CH_3 + Cl^- + HOEt \end{aligned}$$
This product is ethyl but-3-enoate (a terminal, monosubstituted alkene). It is less substituted and it is not conjugated with the carbonyl, so it is thermodynamically less stable.
According to Zaitsev’s rule, “in an elimination the more substituted (and therefore more stable) alkene is formed preferentially.” Furthermore, conjugation with the ester carbonyl provides an additional driving force. Therefore Path 1 overwhelmingly dominates.
Thus the major product is
$$\boxed{CH_3CH = CHCO_2CH_2CH_3}$$
This structure matches Option 3 in the given list.
Hence, the correct answer is Option 3.
Create a FREE account and get:
Educational materials for JEE preparation
