Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The combination of plots which does not represent isothermal expansion of an ideal gas is
We are dealing with an ideal gas that is undergoing an isothermal expansion. By definition an isothermal process takes place at constant temperature, so the absolute temperature $$T$$ does not change while the volume increases and the pressure decreases.
For an ideal gas we always have the ideal-gas equation
$$PV=nRT,$$
where $$n$$ is the amount of substance, $$R$$ is the universal gas constant and $$T$$ is the absolute temperature. During an isothermal process both $$n$$ and $$T$$ remain constant, so their product $$nRT$$ is a single constant, say $$K$$:
$$PV = K. \quad -(1)$$
Equation (1) can be written in several equivalent algebraic ways, each one immediately telling us how one thermodynamic variable depends on another when the temperature is fixed:
• Solving for $$P$$ gives $$P=\dfrac{K}{V}$$ so $$P\propto\dfrac1V.
• Solving for $$V$$ gives $$V=\dfrac{K}{P}$$ so $$V\propto\dfrac1P.
• Solving for $$\dfrac1V$$ gives $$\dfrac1V=\dfrac{P}{K}$$ so $$\dfrac1V\propto P.
• Solving for $$\dfrac1P$$ gives $$\dfrac1P=\dfrac{V}{K}$$ so $$\dfrac1P\propto V.$$
From these proportionalities we can foresee the correct qualitative shapes of the various plots that truly represent an isothermal expansion:
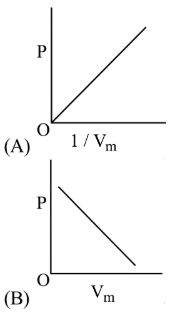
1. A plot of $$P$$ (vertical axis) versus $$V$$ (horizontal axis) must be a rectangular hyperbola that falls continuously as $$V$$ increases. The curve never cuts an axis because neither $$P$$ nor $$V$$ can become zero for a finite temperature.
2. A plot of $$P$$ versus $$\dfrac1V$$ must be a straight line passing through the origin because $$P=K$$\left$$(\dfrac1V$$\right$$).$$ The slope of this straight line is the constant $$K$$.
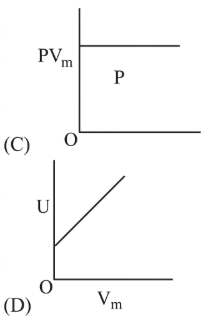
3. A plot of $$V$$ versus $$\dfrac1P$$ must also be a straight line through the origin because $$V=K$$\left$$(\dfrac1P$$\right$$).$$
4. A plot of $$\dfrac1P$$ versus $$\dfrac1V$$ will again be a rectangular hyperbola: multiplying the two plotted variables yields $$$$\left$$(\dfrac{1}{P}$$\right$$)$$\left$$(\dfrac{1}{V}$$\right$$)=\dfrac{1}{K},$$ another constant.
Any diagram that shows a direct proportionality ($$P\propto V$$ or $$\dfrac1P\propto\dfrac1V$$) or a horizontal / vertical line (which would keep either variable fixed) clearly contradicts the inverse relationships listed above, and therefore cannot correspond to an isothermal expansion.
When we inspect the four diagrams supplied in the paper, figures (A), (B), (C) and (D), we observe the following qualitative features:
• Figure (A) depicts a falling rectangular hyperbola in the $$P$$-$$V$$ plane, fully consistent with $$PV=K.
• Figure (B) depicts a straight line with a positive slope in the $$P$$-$$V$$ plane, passing through the origin, i.e. it suggests $$P\propto V,$$ which contradicts Eq. (1).$$
• Figure (C) depicts a straight line through the origin in the $$V$$ versus $$\dfrac1P$$ plane, exactly matching $$V=\dfrac{K}{P}.$$ Hence this is an isothermal graph.
• Figure (D) depicts a straight line with a positive slope in the $$\dfrac1P$$ versus $$\dfrac1V$$ plane, that is $$\dfrac1P\propto\dfrac1V.$$ Multiplying the two axes variables now yields $$$$\left$$(\dfrac{1}{P}$$\right$$)$$\left$$(\dfrac{1}{V}$$\right$$)\propto\dfrac{1}{V}$$\cdot$$\dfrac{1}{V}=\dfrac{1}{V^{2}},$$ which is certainly not the constant $$\dfrac1K$$ required by Eq. (1). So this figure also violates the isothermal condition.
Combining these observations, we see that diagrams (B) and (D) fail to obey the inverse proportionalities mandated by the ideal-gas law at constant temperature, and therefore do not represent an isothermal expansion. The remaining diagrams (A) and (C) obey Eq. (1) and are acceptable.
Among the answer choices provided, option C lists exactly the pair (B) and (D).
Hence, the correct answer is Option C.
Create a FREE account and get:
Educational materials for JEE preparation
