Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The decreasing order of reactivity of the following organic molecules towards AgNO$$_3$$ solution is:
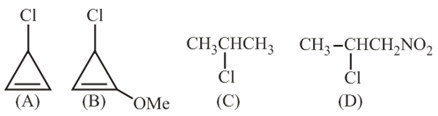
We are asked to arrange the four given halides in the order in which they react with an ethanolic solution of silver nitrate, $$AgNO_3$$. Silver-nitrate in ethanol brings about ionisation of the C-X bond and thus follows an $$S_N1$$ mechanism. In the $$S_N1$$ pathway the rate-determining step is the formation of a carbocation, so the more stable the carbocation, the faster the reaction. Hence we simply have to compare the stabilities of the carbocations that would be produced from the four substrates.
First, let us write the general ionisation step which every substrate must undergo:
$$R-X \;\xrightarrow[\text{EtOH}]{AgNO_3}\; R^{+} \;+\; AgX\!\downarrow$$
The precipitate, $$AgX$$, appears only after the carbocation $$R^{+}$$ has formed, so the relative rate of precipitation mirrors the relative ease of carbocation formation. Now we look at each compound.
Compound (B) possesses a benzylic chloride. When the C-Cl bond breaks, the resulting benzylic carbocation is resonance-stabilised over the aromatic ring. We can draw:
$$\overset{+}{CH_2}-C_6H_5 \;\longleftrightarrow\; CH_2= C_6H_5^{+}$$
Because several equivalent resonance structures distribute the positive charge, the benzylic cation is extremely stable, so (B) will ionise fastest.
Compound (A) is a tertiary alkyl chloride. Tertiary carbocations are stabilised by the +I (electron-releasing) effect and hyperconjugation from three neighbouring alkyl groups. They are very stable, but still slightly less so than the resonance-delocalised benzylic cation. Therefore (A) comes after (B).
Compound (C) is an allylic chloride. Cleavage of the C-Cl bond furnishes an allylic carbocation that is resonance-stabilised over the adjacent C=C bond:
$$\overset{+}{CH_2}-CH=CH_2 \;\longleftrightarrow\; CH_2=CH-CH_2^{+}$$
This resonance gives appreciable stability, but not as much as in a benzylic or tertiary situation, so (C) reacts more slowly than (A).
Compound (D) is a simple primary alkyl chloride without any resonance or strong inductive stabilisation. The primary carbocation generated would be highly unstable, making ionisation very slow. Consequently (D) is the least reactive of the four.
Collecting these observations, the order of decreasing reactivity is
$$\text{(B) > (A) > (C) > (D)}$$
This exactly matches Option 4 in the given list.
Hence, the correct answer is Option 4.
Create a FREE account and get:
Educational materials for JEE preparation
