Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
In the given P-V diagram, a monoatomic gas $$\left(\gamma = \dfrac{5}{3}\right)$$ is first compressed adiabatically from state A to state B. Then it expands isothermally from state B to state C. [Given: $$\left(\dfrac{1}{3}\right)^{0.6} = 0.5$$, ln 2 $$\simeq$$ 0.7].
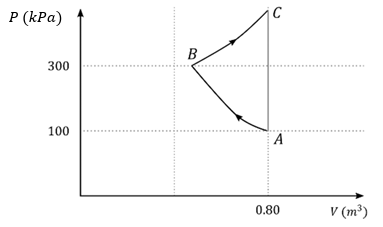
Which of the following statement(s) is(are) correct?
Create a FREE account and get:
Educational materials for JEE preparation
