Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
If the wavelength of the first member of Lyman series of hydrogen is $$\lambda$$. The wavelength of the second member will be
Lyman series: $$\frac{1}{\lambda} = R(1 - 1/n^2)$$.
First member (n=2): $$\frac{1}{\lambda} = R(1-1/4) = \frac{3R}{4}$$.
Second member (n=3): $$\frac{1}{\lambda_2} = R(1-1/9) = \frac{8R}{9}$$.
$$\frac{\lambda_2}{\lambda} = \frac{3R/4}{8R/9} = \frac{27}{32}$$.
$$\lambda_2 = \frac{27}{32}\lambda$$.
The answer is Option (1): $$\frac{27}{32}\lambda$$.
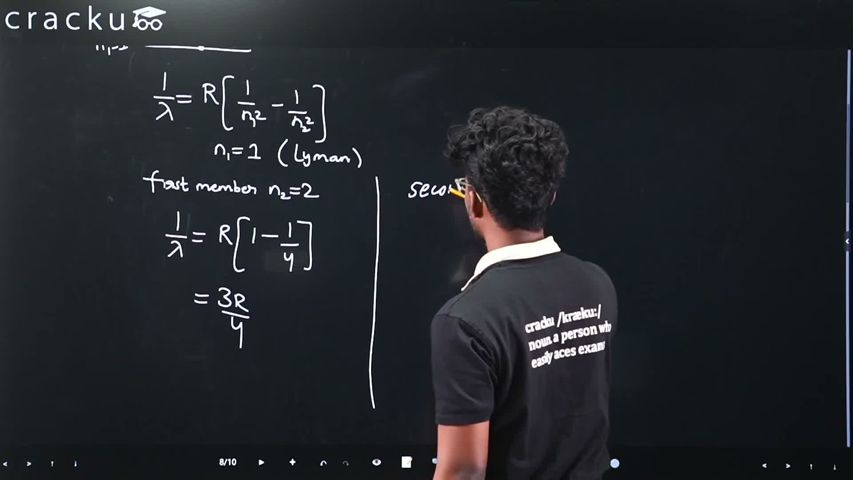
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
