Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $$8.0 \times 10^{-3}$$ m$$^2$$. Initially the gas is at 300 K and occupies a volume of $$2.4 \times 10^{-3}$$ m$$^3$$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by 0.1 m. The force constant of the spring is 8000 N/m and the atmospheric pressure is $$1.0 \times 10^5$$ N/m$$^2$$. The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater. The heat capacity of the heater coil is also negligible).
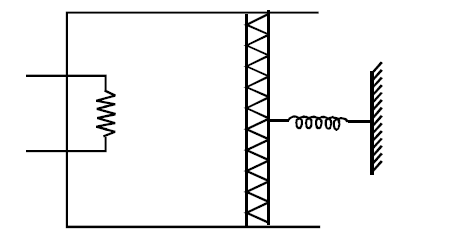
$$V_1 = 2.4 \times 10^{-3} \text{ m}^3,$$
$$P_1 = P_0 = 10^5 \text{ }\frac{\text{N}}{\text{m}^2} \quad \text{and}$$ $$T_1 = 300\text{ K} \qquad \text{(given)}$$
If area of cross-section of piston is $$A$$ and it moves through distance $$x$$ then increment in volume of the gas $$= Ax$$ and if force constant of a spring is $$k$$ then force $$F = kx$$ and pressure $$= F/A = kx/A$$
$$V_2 = V_1 + Ax = 2.4 \times 10^{-3} + 8 \times 10^{-3} \times 0.1 = 3.2 \times 10^{-3}$$
$$\text{and } P_2 = P_0 + \frac{kx}{A} = 10^5 + \frac{8000 \times 0.1}{8 \times 10^{-3}} = 2 \times 10^5$$
$$\text{From ideal gas equation } \frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2}$$
$$\implies \frac{10^5 \times 2.4 \times 10^{-3}}{300} = \frac{2 \times 10^5 \times 3.2 \times 10^{-3}}{T_2}$$
$$\implies T_2 = 800\text{ K}$$
Create a FREE account and get:
Educational materials for JEE preparation
