Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
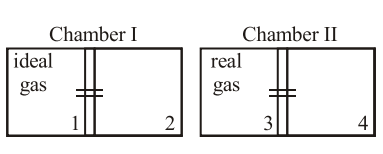
There are two identical chambers, completely thermally insulated from surroundings. Both chambers have a partition wall dividing the chambers in two compartments. Compartment 1 is filled with an ideal gas and Compartment 3 is filled with a real gas. Compartments 2 and 4 are vacuum. A small hole (orifice) is made in the partition walls and the gases are allowed to expand in vacuum.
Statement - 1 : No change in the temperature of the gas takes place when ideal gas expands in vacuum. However, the temperature of real gas goes down (cooling) when it expands in vacuum.
Statement - 2 : The internal energy of an ideal gas is only kinetic. The internal energy of a real gas is kinetic as well as potential.
The temperature of a real gas does not always decrease during free expansion. Depending on the gas and conditions, it may increase, decrease, or remain unchanged.
In an ideal gas, intermolecular forces are neglected, so the internal energy does not contain intermolecular potential energy. Hence, the internal energy of an ideal gas depends only on molecular kinetic energy (translational, rotational, and vibrational depending on the molecule and temperature). For a real gas, intermolecular forces exist, so its internal energy contains both kinetic and potential energy components.
Create a FREE account and get:
Educational materials for JEE preparation
