NTA JEE Main 9th April 2013 Online
For the following questions answer them individually
NTA JEE Main 9th April 2013 Online - Question 1
From the following, the quantity (constructed from the basic constants of nature), that has the dimensions, as well as correct order of magnitude, vis-a-vis typical atomic size, is:
NTA JEE Main 9th April 2013 Online - Question 2
Two springs of force constants 300 N/m (Spring A) and 400 N/m (Spring B) are joined together in series. The combination is compressed by 8.75 cm. The ratio of energy stored in A and B is $$\frac{E_A}{E_B}$$. Then $$\frac{E_A}{E_B}$$ is equal to:
NTA JEE Main 9th April 2013 Online - Question 3
A bullet of mass 10 g and speed 500 m/s is fired into a door and gets embedded exactly at the centre of the door. The door is 1.0 m wide and weighs 12 kg. It is hinged at one end and rotates about a vertical axis practically without friction. The angular speed of the door just after the bullet embeds into it will be :
NTA JEE Main 9th April 2013 Online - Question 4
A uniform sphere of weight W and radius 5 cm is being held by a string as shown in the figure. The tension in the string will be :
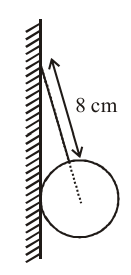
NTA JEE Main 9th April 2013 Online - Question 5
The gravitational field, due to the 'left over part' of a uniform sphere (from which a part as shown, has been 'removed out'), at a very far off point, P, located as shown, would be (nearly) :
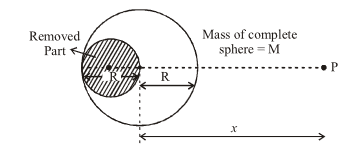
NTA JEE Main 9th April 2013 Online - Question 6
If the ratio of lengths, radii and Young's moduli of steel and brass wires in the figure are a, b and c respectively, then the corresponding ratio of increase in their lengths is :
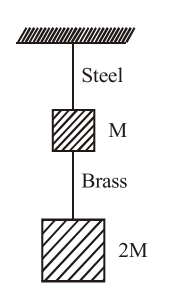
NTA JEE Main 9th April 2013 Online - Question 7
This question has Statement-1 and Statement-2. Of the four choices given after the Statements, choose the one that best describes the two Statements.
Statement-1: A capillary is dipped in a liquid and liquid rises to a height h in it. As the temperature of the liquid is raised, the height h increases (if the density of the liquid and the angle of contact remain the same).
Statement-2: Surface tension of a liquid decreases with the rise in its temperature.
NTA JEE Main 9th April 2013 Online - Question 8
On a linear temperature scale Y, water freezes at $$-160°$$Y and boils at $$-50°$$Y. On this Y scale, a temperature of 340 K would be read as : (water freezes at 273 K and boils at 373 K)
NTA JEE Main 9th April 2013 Online - Question 9
Figure shows the variation in temperature ($$\Delta T$$) with the amount of heat supplied (Q) in an isobaric process corresponding to a monoatomic (M), diatomic (D) and a polyatomic (P) gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines a, b and c respectively correspond to :
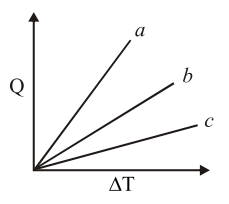
NTA JEE Main 9th April 2013 Online - Question 10
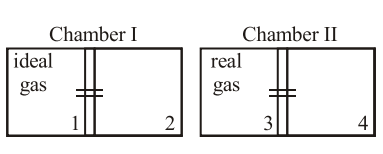
There are two identical chambers, completely thermally insulated from surroundings. Both chambers have a partition wall dividing the chambers in two compartments. Compartment 1 is filled with an ideal gas and Compartment 3 is filled with a real gas. Compartments 2 and 4 are vacuum. A small hole (orifice) is made in the partition walls and the gases are allowed to expand in vacuum.
Statement - 1 : No change in the temperature of the gas takes place when ideal gas expands in vacuum. However, the temperature of real gas goes down (cooling) when it expands in vacuum.
Statement - 2 : The internal energy of an ideal gas is only kinetic. The internal energy of a real gas is kinetic as well as potential.
.webp)


