Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
An engine operates by taking $$n$$ moles of an ideal gas through the cycle $$ABCDA$$ shown in figure. The thermal efficiency of the engine is: (Take $$C_v = 1.5R$$, where $$R$$ is gas constant)
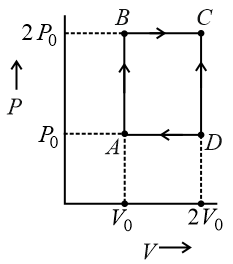
Let us read the diagram (not to scale) as a rectangle in the $$P{-}V$$ plane whose corners are
$$A(P_0,V_0)$$, $$B(2P_0,V_0)$$, $$C(2P_0,2V_0)$$, $$D(P_0,2V_0)$$.
Thus
AB is an isochoric rise in pressure, BC is an isobaric expansion, CD is an isochoric fall in pressure and DA is an isobaric compression, finally returning to the initial state. The working substance is an ideal gas containing $$n$$ moles with $$C_v = 1.5R$$, so that $$C_p = C_v + R = 2.5R.$$ We now follow the gas round the cycle, calculating the work $$W$$ and the heat absorbed $$Q_{\text{in}}$$ step by step.
State-variables at the four corners
The ideal-gas equation $$PV = nRT$$ gives the temperatures
$$T_A = \frac{P_0 V_0}{nR},\qquad T_B = \frac{2P_0 V_0}{nR}=2T_A,\qquad T_C = \frac{2P_0\,(2V_0)}{nR}=4T_A,\qquad T_D = \frac{P_0\,(2V_0)}{nR}=2T_A.$$
Process AB (isochoric, $$V=V_0$$)
Work done: $$W_{AB}=0.$$
Change in internal energy: $$\Delta U_{AB}=nC_v\,(T_B-T_A)=nC_v(2T_A-T_A)=nC_vT_A.$$
Heat absorbed: $$Q_{AB}=W_{AB}+\Delta U_{AB}=nC_vT_A.$$
Process BC (isobaric, $$P=2P_0$$)
Work done: $$W_{BC}=P\,(V_2-V_1)=2P_0\,(2V_0-V_0)=2P_0V_0.$$
Convert the above work into a more useful form by replacing $$P_0V_0$$ with $$nRT_A$$ obtained from $$T_A$$:
$$W_{BC}=2\,nRT_A.$$
Change in internal energy: $$\Delta U_{BC}=nC_v\,(T_C-T_B)=nC_v\,(4T_A-2T_A)=2nC_vT_A.$$
Heat absorbed: $$Q_{BC}=W_{BC}+\Delta U_{BC}=2nRT_A+2nC_vT_A=2n(C_v+R)T_A=2nC_pT_A.$$
Process CD (isochoric cooling)
This branch rejects heat, so its contribution is not counted in $$Q_{\text{in}}.$$
Process DA (isobaric compression)
This branch also rejects heat and therefore does not enter $$Q_{\text{in}}.$$
Total heat absorbed during the cycle
$$Q_{\text{in}} = Q_{AB} + Q_{BC} = nC_vT_A + 2nC_pT_A = nT_A\,(C_v + 2C_p).$$
Insert the numerical values $$C_v = 1.5R,\; C_p = 2.5R$$:
$$Q_{\text{in}} = nT_A\,[\,1.5R + 2(2.5R)\,] = nT_A(1.5R + 5R)=6.5\,nRT_A.$$
Net work done per cycle
The work done is the area of the rectangle $$ABCDA$$:
$$W = (P_C - P_D)\,(V_C - V_B) = (2P_0 - P_0)\,(2V_0 - V_0)=P_0V_0.$$
Replacing $$P_0V_0$$ by $$nRT_A$$ again gives
$$W = nRT_A.$$
Thermal efficiency
The definition is $$\eta = \dfrac{W}{Q_{\text{in}}}.$$ Substituting the expressions just obtained,
$$\eta = \frac{nRT_A}{6.5\,nRT_A}= \frac1{6.5}\approx 0.154\simeq 0.15.$$
Hence, the correct answer is Option B.
Create a FREE account and get:
Educational materials for JEE preparation
