NTA JEE Mains 6th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 6th April 2024 Shift 1 - Question 51
Frequency of the de-Broglie wave of electron in Bohr's first orbit of hydrogen atom is _______ $$\times 10^{13}$$ Hz (nearest integer). [Given : $$R_H$$ (Rydberg constant) $$= 2.18 \times 10^{-18}$$ J, $$h$$ (Planck's constant) $$= 6.6 \times 10^{-34}$$ J.s.]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 52
Number of molecules from the following which can exhibit hydrogen bonding is _______ (nearest integer):
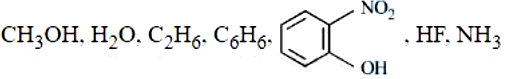
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 53
An ideal gas, $$\bar{C}_v = \frac{5}{2}R$$, is expanded adiabatically against a constant pressure of 1 atm until it doubles in volume. If the initial temperature and pressure is $$298 \text{ K}$$ and $$5 \text{ atm}$$, respectively then the final temperature is _______ K (nearest integer). [$$\bar{C}_v$$ is the molar heat capacity at constant volume]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 54
The major product of the following reaction is $$P$$. $$CH_3C \equiv C-CH_3 \xrightarrow[\text{(ii) dil. KMnO}_4, 273\text{ K}]{\text{(i) Na/liq. NH}_3} P$$. Number of oxygen atoms present in product '$$P$$' is _______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 55
Consider the dissociation of the weak acid HX as given below $$HX(aq) \rightleftharpoons H^+(aq) + X^-(aq)$$, $$K_a = 1.2 \times 10^{-5}$$ [$$K_a$$ : dissociation constant]. The osmotic pressure of $$0.03$$ M aqueous solution of HX at $$300 \text{ K}$$ is _______ $$\times 10^{-2}$$ bar (nearest integer). [Given : $$R = 0.083 \text{ L bar mol}^{-1} \text{K}^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 56
Time required for 99.9% completion of a first order reaction is _______ times the time required for completion of 90% reaction. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 57
Among $$CrO$$, $$Cr_2O_3$$ and $$CrO_3$$, the sum of spin-only magnetic moment values of basic and amphoteric oxides is _______ $$\times 10^{-2}$$ BM (nearest integer). (Given atomic number of Cr is 24)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 58
The difference in the 'spin-only' magnetic moment values of $$KMnO_4$$ and the manganese product formed during titration of $$KMnO_4$$ against oxalic acid in acidic medium is _______ BM. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 59
The major products from the following reaction sequence are product A and product B. The total sum of $$\pi$$ electrons in product A and product B are _______ (nearest integer)
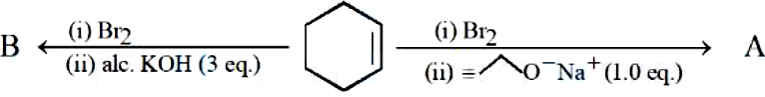
789
456
123
0.-
Clear All
NTA JEE Mains 6th April 2024 Shift 1 - Question 60
$$9.3 \text{ g}$$ of pure aniline upon diazotisation followed by coupling with phenol gives an orange dye. The mass of orange dye produced (assume 100% yield/conversion) is _______ g. (nearest integer)
789
456
123
0.-
Clear All


