NTA JEE Mains 6th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 6th April 2024 Shift 1 - Question 41
Given below are two statements: Statement I : Gallium is used in the manufacturing of thermometers. Statement II : A thermometer containing gallium is useful for measuring the freezing point of brine solution (256 K). In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Question 42
A conductivity cell with two electrodes (dark side) are half filled with infinitely dilute aqueous solution of a weak electrolyte. If volume is doubled by adding more water at constant temperature, the molar conductivity of the cell will -
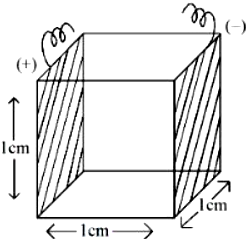
NTA JEE Mains 6th April 2024 Shift 1 - Question 43
The number of elements from the following that do not belong to lanthanoids is $$Eu, Cm, Er, Tb, Yb$$ and $$Lu$$
NTA JEE Mains 6th April 2024 Shift 1 - Question 44
Match List I with List II
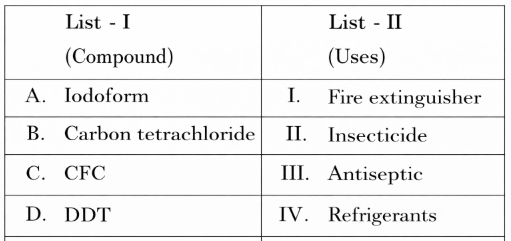
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Question 45
The following complexes $$[CoCl(NH_3)_5]^{2+}$$ (A), $$[Co(CN)_6]^{3-}$$ (B), $$[Co(NH_3)_5(H_2O)]^{3+}$$ (C), $$[Cu(H_2O)_4]^{2+}$$ (D). The correct order of A, B, C and D in terms of wavenumber of light absorbed is :
NTA JEE Mains 6th April 2024 Shift 1 - Question 46
Given below are two statements : Statement I : Picric acid is 2,4,6 - trinitrotoluene. Statement II : Phenol - 2,4 - disulphonic acid is treated with Conc. $$HNO_3$$ to get picric acid. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 6th April 2024 Shift 1 - Question 47
In Reimer - Tiemann reaction, phenol is converted into salicylaldehyde through an intermediate. The structure of intermediate is _____
NTA JEE Mains 6th April 2024 Shift 1 - Question 48
Which among the following aldehydes is most reactive towards nucleophilic addition reactions?
NTA JEE Mains 6th April 2024 Shift 1 - Question 49
Match List I with List II
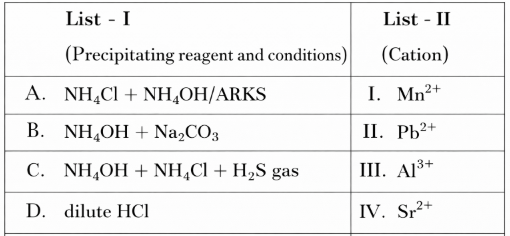
Choose the correct answer from the options given below:
NTA JEE Mains 6th April 2024 Shift 1 - Question 50
DNA molecule contains 4 bases whose structures are shown below. One of the structures is not correct, identify the incorrect base structure.


