NTA JEE Mains 5th April 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 5th April 2024 Shift 1 - Question 51
$$9.3 \text{ g}$$ of pure aniline is treated with bromine water at room temperature to give a white precipitate of the product '$$P$$'. The mass of product '$$P$$' obtained is $$26.4 \text{ g}$$. The percentage yield is ______ %.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 52
The value of Rydberg constant $$(R_H)$$ is $$2.18 \times 10^{-18} \text{ J}$$. The velocity of electron having mass $$9.1 \times 10^{-31} \text{ kg}$$ in Bohr's first orbit of hydrogen atom $$=$$ ______ $$\times 10^5 \text{ ms}^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 53
In the Lewis dot structure for $$NO_2^-$$, total number of valence electrons around nitrogen is ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 54
The heat of combustion of solid benzoic acid at constant volume is $$-321.30 \text{ kJ}$$ at $$27°C$$. The heat of combustion at constant pressure is $$(-321.30 - xR) \text{ kJ}$$, the value of $$x$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 55
An artificial cell is made by encapsulating $$0.2 \text{ M}$$ glucose solution within a semipermeable membrane. The osmotic pressure developed when the artificial cell is placed within a $$0.05 \text{ M NaCl}$$ solution at $$300 \text{ K}$$ is ______ $$\times 10^{-1} \text{ bar}$$. (nearest integer). [Given : $$R = 0.083 \text{ L bar mol}^{-1} \text{ K}^{-1}$$] Assume complete dissociation of $$NaCl$$
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 56
During Kinetic study of reaction $$2A + B \to C + D$$, the following results were obtained :
I: A = 0.1 M, B = 0.1 M, initial rate of formation of D = $$6.0 \times 10^{-3}$$
II: A = 0.3 M, B = 0.2 M, initial rate = $$7.20 \times 10^{-2}$$
III: A = 0.3 M, B = 0.4 M, initial rate = $$2.88 \times 10^{-1}$$
IV: A = 0.4 M, B = 0.1 M, initial rate = $$2.40 \times 10^{-2}$$
Based on above data, overall order of the reaction is ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 57
The spin-only magnetic moment value of the ion among $$Ti^{2+}, V^{2+}, Co^{3+}$$ and $$Cr^{2+}$$, that acts as strong oxidising agent in aqueous solution is ______ BM (Near integer). (Given atomic numbers : $$Ti : 22, V : 23, Cr : 24, Co : 27$$)
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 58
The number of halobenzenes from the following that can be prepared by Sandmeyer's reaction is ______
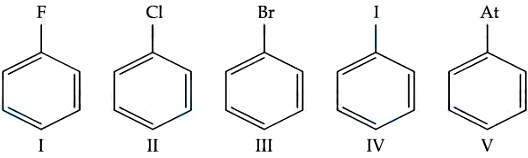
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 59
Consider the given chemical reaction sequence :
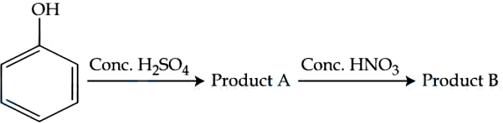
Total sum of oxygen atoms in Product A and Product B are ______
789
456
123
0.-
Clear All
NTA JEE Mains 5th April 2024 Shift 1 - Question 60
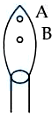
In a borax bead test under hot condition, a metal salt (one from the given) is heated at point B of the flame, resulted in green colour salt bead. The spin-only magnetic moment value of the salt is ______ BM (Nearest integer) [Given atomic number of $$Cu = 29, Ni = 28, Mn = 25, Fe = 26$$]
789
456
123
0.-
Clear All


