NTA JEE Main 28th July 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 31
Which of the following pair is not isoelectronic species?
(Atomic numbers Ho=67; Er=68; Yb=70; Lu=71; Eu=63; Tb=65; Tm=69)
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 32
The correct decreasing order for metallic character is
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 33
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Zero orbital overlap is an out of phase overlap.
Reason R: It results due to different orientation/direction of approach of orbitals.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 34
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R
Assertion A: The reduction of a metal oxide is easier if the metal formed is in liquid state than solid state.
Reason R: The value of $$\Delta G^\theta$$ becomes more on negative side as entropy is higher in liquid state than solid state.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 35
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Permanganate titrations are not performed in presence of hydrochloric acid.
Reason R: Chlorine is formed as a consequence of oxidation of hydrochloric acid.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 36
The products obtained during treatment of hard water using Clark's method are
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 37
Statement I: An alloy of lithium and magnesium is used to make aircraft plates.
Statement II: The magnesium ions are important for cell-membrane integrity.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 38
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Thin layer chromatography is an adsorption chromatography.
Reason R: A thin layer of silica gel is spread over a glass plate of suitable size in thin layer chromatography which acts as an adsorbent.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 39
Arrange the following in increasing order of reactivity towards nitration
A. p-xylene
B. bromobenzene
C. mesitylene
D. nitrobenzene
E. benzene
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 40
White phosphorus reacts with thionyl chloride to give
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 41
Concentrated $$HNO_3$$ reacts with Iodine to give
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 42
Dinitrogen and dioxygen the main constituents of air do not react with each other in atmosphere to form oxides of nitrogen because
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 43
Match List-I with List-II
| List-I (Complex) | List-II (Hybridization) |
|---|---|
| A. $$Ni(CO)_4$$ | I. $$sp^3$$ |
| B. $$[Ni(CN)_4]^{2-}$$ | II. $$sp^3d^2$$ |
| C. $$[Co(CN)_6]^{3-}$$ | III. $$d^2sp^3$$ |
| D. $$[CoF_6]^{3-}$$ | IV. $$dsp^2$$ |
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 44
The major product in the given reaction is
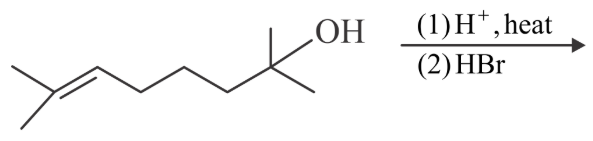
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 45
Compound I is heated with Conc. HI to give a hydroxy compound A which is further heated with Zn dust to give compound B. Identify A and B.
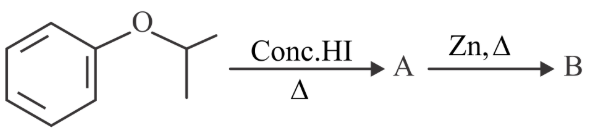
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 46
Find out the major product for the above reaction.
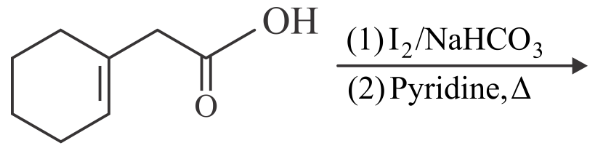
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 47
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Aniline on nitration yields ortho, meta & para nitro derivatives of aniline.
Reason R: Nitrating mixture is a strong acidic mixture.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 48
Match List I with List II
| List-I (Polymer) | List-II (Nature) |
|---|---|
A
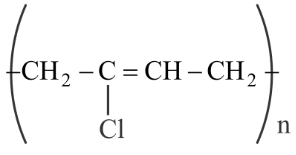 | I. Thermosetting polymer |
B
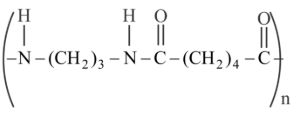 | II. Fibers |
C
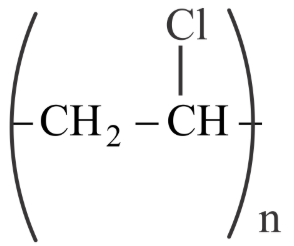 | III. Elastomer |
D
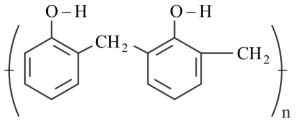 | IV. Thermoplastic polymer |
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 49
Two statements in respect of drug-enzyme interaction are given below
Statement I: Action of an enzyme can be blocked only when an inhibitor blocks the active site of the enzyme.
Statement II: An inhibitor can form a strong covalent bond with the enzyme.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 50
The formulas of A and B for the following reaction sequence are
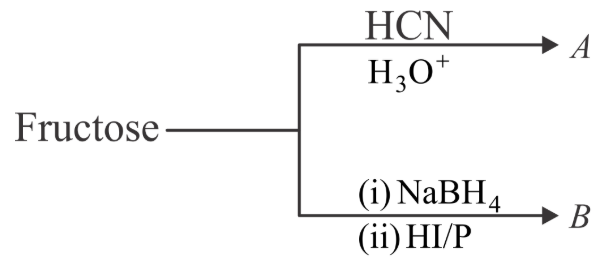
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 51
2 L of 0.2 M $$H_2SO_4$$ is reacted with 2 L of 0.1 M NaOH solution, the molarity of the resulting product $$Na_2SO_4$$ in the solution is _____ millimolar.
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 52
If the wavelength for an electron emitted from $$H^-$$ atom is $$3.3 \times 10^{-10}$$ m, then energy absorbed by the electron in its ground state compared to minimum energy required for its escape from the atom, is _____ times. [Given: $$h = 6.626 \times 10^{-34}$$ Js, Mass of electron $$= 9.1 \times 10^{-31}$$ kg]
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 53
Among the following the number of state variable is
Internal energy (U), Volume (V), Heat (q), Enthalpy (H)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 54
At 600 K, 2 mol of NO are mixed with 1 mol of $$O_2$$.
$$2NO(g) + O_2(g) \rightleftharpoons 2NO_2(g)$$
The reaction occurring as comes to equilibrium under a total pressure of 1 atm. Analysis of the system shows that 0.6 mol of oxygen are present at equilibrium. The equilibrium constant for the reaction is _____
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 55
On reaction with stronger oxidizing agent like $$KIO_4$$, hydrogen peroxide oxidizes with the evolution of $$O_2$$. The oxidation number of I in $$KIO_4$$ changes to
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 56
A sample of 0.125 g of an organic compound when analysed by Duma's method yields 22.78 mL of nitrogen gas collected over KOH solution at 280 K and 759 mmHg. The percentage of nitrogen in the given organic compound is _____
(a) The vapour pressure of water at 280 K is 14.2 mmHg
(b) $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 57
Metal M crystallizes into a FCC lattice with the edge length of $$4.0 \times 10^{-8}$$ cm. The atomic mass of the metal is _____ g/mol. (Use: $$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$, density of metal, $$M = 9.03$$ g cm$$^{-3}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 58
A gaseous mixture of two substances A and B, under a total pressure of 0.8 atm is in equilibrium with an ideal liquid solution. The mole fraction of substance A is 0.5 in the vapour phase and 0.2 in the liquid phase. The vapour pressure of pure liquid A is _____ atm. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 59
For a reaction, given below is the graph of $$\ln k$$ vs $$\frac{1}{T}$$. The activation energy for the reaction is equal to _____ cal mol$$^{-1}$$. (Given: $$R = 2$$ cal K$$^{-1}$$ mol$$^{-1}$$)
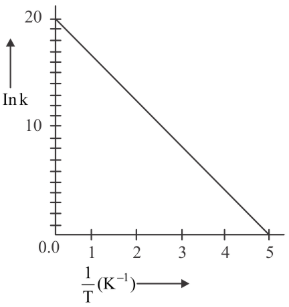
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Chemistry - Question 60
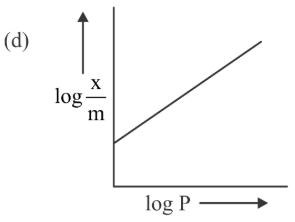
Among the following the number of curves not in accordance with Freundlich adsorption isotherm is
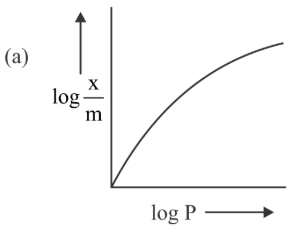
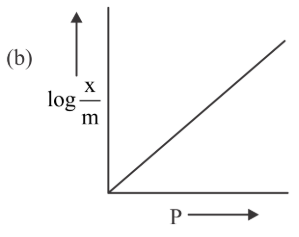
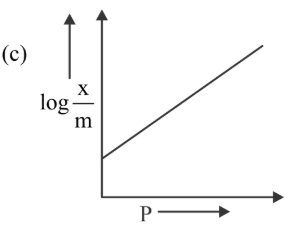
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




