NTA JEE Main 28th July 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 31
Identify the incorrect statement from the following.
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 32
In which of the following pairs, electron gain enthalpies of constituent elements are nearly the same or identical?
(A) Rb and Cs
(B) Na and K
(C) Ar and Kr
(D) I and At
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 33
Which of the following relation is not correct?
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 34
The metal salts formed during softening of hard water using Clark's method are
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 35
For kinetic study of the reaction of iodide ion with $$H_2O_2$$ at room temperature:
(A) Always use freshly prepared starch solution.
(B) Always keep the concentration of sodium thiosulphate solution less than that of KI solution.
(C) Record the time immediately after the appearance of blue colour.
(D) Record the time immediately before the appearance of blue colour.
(E) Always keep the concentration of sodium thiosulphate solution more than that of KI solution.
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 36
Which of the following statement is incorrect?
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 37
Match List-I with List-II.
| List-I | List-II |
|---|---|
A
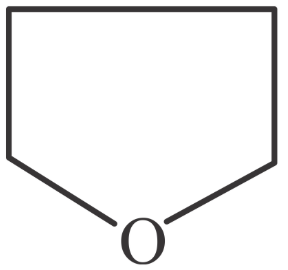 | I. Spiro compound |
B
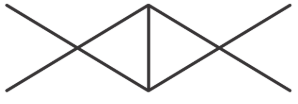 | II. Aromatic compound |
C
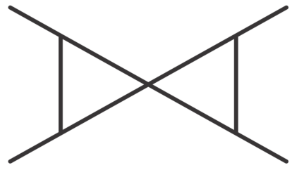 | III. Non-planar Heterocyclic compound |
D
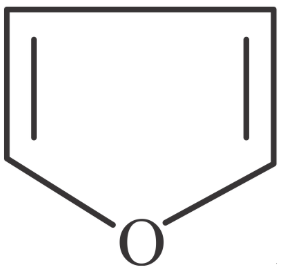 | IV. Bicyclo compound |
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 38
Among the following marked proton of which compound shows lowest pKa value?
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 39
Choose the correct option for the following reactions.
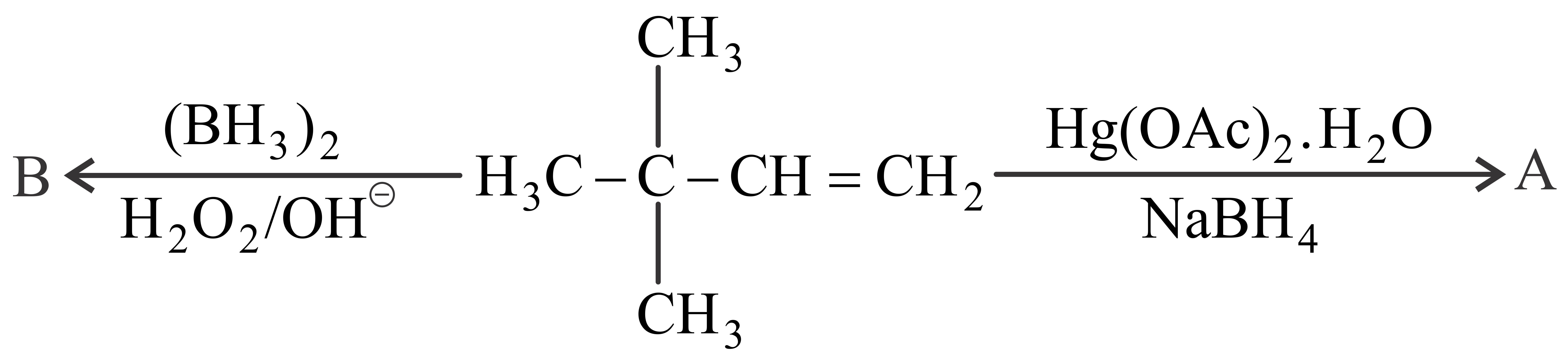
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 40
Identify the correct statement for the below given transformation.
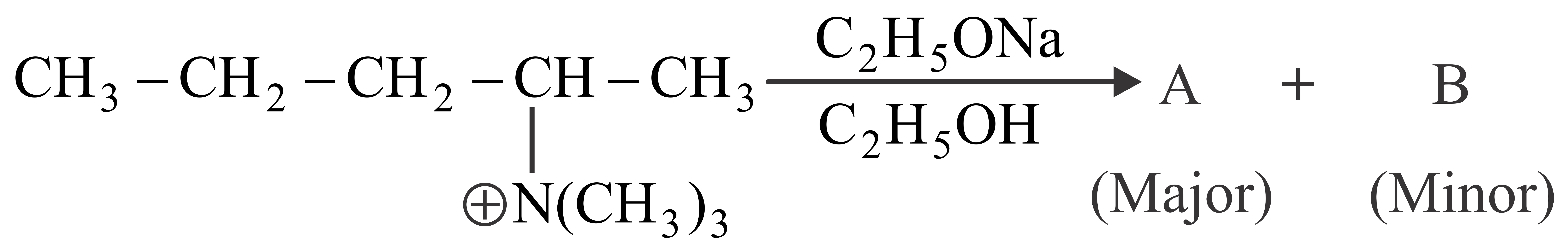
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 41
Given below are two statements:
Statement I: In polluted water values of both dissolved oxygen and BOD are very low.
Statement II: Eutrophication results in decrease in the amount of dissolved oxygen.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 42
Match List-I with List-II.
| List-I | List-II |
|---|---|
| A. $$Cd(s) + 2Ni(OH)_3(s) \to CdO(s) + 2Ni(OH)_2(s) + H_2O(l)$$ | I. Primary battery |
| B. $$Zn(Hg) + HgO(s) \to ZnO(s) + Hg(l)$$ | II. Discharging of secondary battery |
| C. $$2PbSO_4(s) + 2H_2O(l) \to Pb(s) + PbO_2(s) + 2H_2SO_4(aq)$$ | III. Fuel cell |
| D. $$2H_2(g) + O_2(g) \to 2H_2O(l)$$ | IV. Charging of secondary battery |
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 43
Which of the reaction is suitable for concentrating ore by leaching process?
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 44
Match List-I with List-II.
| List-I | List-II |
|---|---|
| A. $$4NH_3(g) + 5O_2(g) \to 4NO(g) + 6H_2O(g)$$ | I. NO(g) |
| B. $$N_2(g) + 3H_2(g) \to 2NH_3(g)$$ | II. $$H_2SO_4(l)$$ |
| C. $$C_{12}H_{22}O_{11}(aq) + H_2O(l) \to C_6H_{12}O_6 (Glucose) + C_6H_{12}O_6 (Fructose)$$ | III. Pt(s) |
| D. $$2SO_2(g) + O_2(g) \to 2SO_3(g)$$ | IV. Fe(s) |
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 45
Match List-I with List-II, match the gas evolved during each reaction.
| List-I | List-II |
|---|---|
| A. $$(NH_4)_2Cr_2O_7 \xrightarrow{\Delta}$$ | I. $$H_2$$ |
| B. $$KMnO_4 + HCl \to$$ | II. $$N_2$$ |
| C. $$Al + NaOH + H_2O \to$$ | III. $$O_2$$ |
| D. $$NaNO_3 \xrightarrow{\Delta}$$ | IV. $$Cl_2$$ |
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 46
Which of the following has least tendency to liberate $$H_2$$ from mineral acids?
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 47
Identify the major product A and B for the below given reaction sequence.
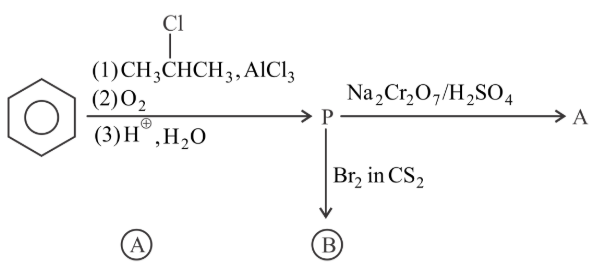
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 48
Terylene polymer is obtained by condensation of
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 49
Statements about Enzyme Inhibitor Drugs are given below:
(A) There are Competitive and Non-competitive inhibitor drugs.
(B) These can bind at the active sites and allosteric sites.
(C) Competitive Drugs are allosteric site blocking drugs.
(D) Non-competitive Drugs are active site blocking drugs.
Choose the correct answer from the options given below:
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 50
For the below given cyclic hemiacetal X, the correct pyranose structure is
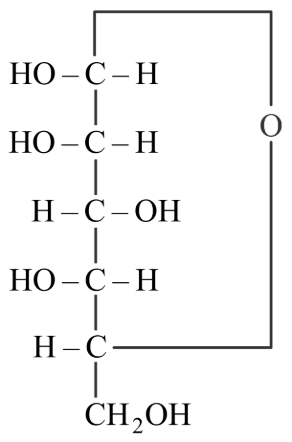
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 51
In the given reaction, $$X + Y + 3Z \rightleftharpoons XYZ_3$$. If one mole of each of X and Y with 0.05 mol of Z gives compound $$XYZ_3$$. (Given: Atomic masses of X, Y and Z are 10, 20 and 30 amu, respectively). The yield of $$XYZ_3$$ is _____ g.
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 52
On complete combustion of 0.492 g of an organic compound containing C, H and O, 0.7938 g of $$CO_2$$ and 0.4428 g of $$H_2O$$ was produced. The % composition of oxygen in the compound is _____ (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 53
The number of paramagnetic species among the following is $$B_2, Li_2, C_2, C_2^-, O_2^{2-}, O_2^+$$ and $$He_2^+$$
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 54
$$K_a$$ for butyric acid ($$C_3H_7COOH$$) is $$2 \times 10^{-5}$$. The pH of 0.2 M solution of butyric acid is _____ $$\times 10^{-1}$$. (Nearest integer) [Given $$\log 2 = 0.30$$]
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 55
An element M crystallises in a body centred cubic unit cell with a cell edge of 300 pm. The density of the element is 6.0 g cm$$^{-3}$$. The number of atoms present in 180 g of the element is _____ $$\times 10^{23}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 56
150 g of acetic acid was contaminated with 10.2 g ascorbic acid $$(C_6H_8O_6)$$ to lower down its freezing point by $$x \times 10^{-1}$$ °C. The value of $$x$$ is _____ (Nearest integer). [Given $$K_f = 3.9$$ K kg mol$$^{-1}$$; Molar mass of ascorbic acid = 176 g mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 57
For the given first order reaction $$A \to B$$ the half life of the reaction is 0.3010 min. The ratio of the initial concentration of reactant to the concentration of reactant at time 2.0 min will be equal to _____. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 58
The number of inter halogens from the following having square pyramidal structure is
$$ClF_3, IF_7, BrF_5, BrF_3, I_2Cl_6, IF_5, ClF, ClF_5$$
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 59
The disproportionation of $$MnO_4^{2-}$$ in acidic medium resulted in the formation of two manganese compounds A and B. If the oxidation state of Mn in B is smaller than that of A, then the spin-only magnetic moment $$\mu$$ value of B in BM is _____ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 1 - Chemistry - Question 60
Total number of relatively more stable isomer(s) possible for octahedral complex $$[Cu(en)_2(SCN)_2]$$ will be _____
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




