NTA JEE Main 26th June 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 31
The number of radial and angular nodes in $$4d$$ orbital are, respectively
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 32
Which of the following is a metalloid?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 33
The oxide which contains an odd electron at the nitrogen atom is
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 34
Which one of the following is an example of disproportionation reaction?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 35
Boiling of hard water is helpful in removing the temporary hardness by converting calcium hydrogen carbonate and magnesium hydrogen carbonate to
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 36
s-block element which cannot be qualitatively confirmed by the flame test is
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 37
The correct order of nucleophilicity is
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 38
The major product of the following reaction is:
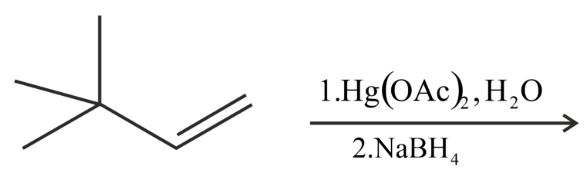
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 39
Halogenation of which one of the following will yield m-substituted product with respect to methyl group as a major product?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 40
The measured BOD values for four different water samples (A - D) are as follows: A = 3 ppm; B = 18 ppm; C = 21 ppm; D = 4 ppm. The water samples which can be called as highly polluted with organic wastes, are
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 41
The role of depressants in 'Froth Floation method' is to
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 42
The most common oxidation state of Lanthanoid elements is $$+3$$. Which of the following is likely to deviate easily from $$+3$$ oxidation state?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 43
Toluene can be easily converted into benzaldehyde by which of the following reagents?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 44
The final product 'A' in the following reaction sequence
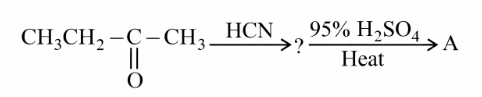
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 45
The reagent, from the following, which converts benzoic acid to benzaldehyde in one step is
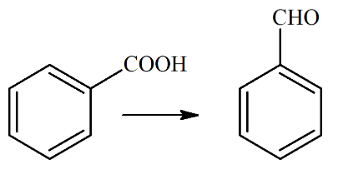
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 46
Which statement is NOT correct for p-toluenesulphonyl chloride?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 47
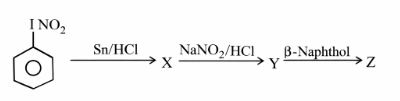
Identify 'Z' among the following
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 48
Match List I with List II.
| List-I (Enzyme) | List-II (Conversion of) |
|---|---|
| A. Invertase | I. Starch into maltose |
| B. Zymase | II. Maltose into glucose |
| C. Diastase | III. Glucose into ethanol |
| D. Maltase | IV. Cane sugar into glucose |
Choose the most appropriate answer from the options given below
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 49
Which of the following is NOT an example of synthetic detergent?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 50
Which one of the following is a water soluble vitamin, that is not excreted easily?
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 51
CNG is an important transportation fuel. When $$100$$ g CNG is mixed with $$208$$ g oxygen in vehicles, it leads to the formation of $$CO_2$$ and $$H_2O$$ and produces large quantity of heat during this combustion, then the amount of carbon dioxide, produced in grams is ______ [nearest integer] [Assume CNG to be methane]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 52
The moles of methane required to produce $$81$$ g of water after complete combustion is ______ $$\times 10^{-2}$$ mol. [nearest integer]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 53
Amongst $$SF_4, XeF_4, CF_4$$ and $$H_2O$$, the number of species with two lone pairs of electrons is ______
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 54
A fish swimming in water body when taken out from the water body is covered with a film of water of weight $$36$$ g. When it is subjected to cooking at $$100°$$C, then the internal energy for vaporization in kJ mol$$^{-1}$$ is ______ [integer]
[Assume steam to be an ideal gas. Given $$\Delta_{vap}H^\circ$$ for water at $$373$$ K and $$1$$ bar is $$41.1$$ kJ mol$$^{-1}$$ : $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 55
$$40\%$$ of HI undergoes decomposition to $$H_2$$ and $$I_2$$ at $$300$$ K. $$\Delta G^\circ$$ for this decomposition reaction at one atmosphere pressure is ______ J mol$$^{-1}$$ [nearest integer]
(Use $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$; $$\log 2 = 0.3010$$, $$\ln 10 = 2.3$$, $$\log 3 = 0.477$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 56
In a solid AB, A atoms are in ccp arrangement and B atoms occupy all the octahedral sites. If two atoms from the opposite faces are removed, then the resultant stoichiometry of the compound is $$A_xB_y$$. The value of $$x$$ is ______ [nearest integer]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 57
The osmotic pressure exerted by a solution prepared by dissolving $$2.0$$ g of protein of molar mass $$60$$ kg mol$$^{-1}$$ in $$200$$ mL of water at $$27°$$C is ______ Pa. [integer value] (use $$R = 0.083$$ L bar mol$$^{-1}$$ K$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 58
$$Cu(s) + Sn^{2+}(0.001M) \to Cu^{2+}(0.01M) + Sn(s)$$
The Gibbs free energy change for the above reaction at $$298$$ K is $$x \times 10^{-1}$$ kJ mol$$^{-1}$$. The value of $$x$$ is ______ [nearest integer]
[Given : $$E^\circ_{Cu^{2+}/Cu} = 0.34$$ V; $$E^\circ_{Sn^{2+}/Sn} = -0.14$$ V; $$F = 96500$$ C mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 59
Catalyst A reduces the activation energy for a reaction by $$10$$ kJ mol$$^{-1}$$ at $$300$$ K. The ratio of rate constants, $$\frac{k_T \text{ Catalysed}}{k_T \text{ Uncatalysed}}$$ is $$e^x$$. The value of $$x$$ is ______ [nearest integer] [Assume that the pre-exponential factor is same in both the cases. Given $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th June 2022 Shift 2 - Chemistry - Question 60
Reaction of $$[Co(H_2O)_6]^{2+}$$ with excess ammonia and in the presence of oxygen results into a diamagnetic product. Number of electrons present in $$t_{2g}$$-orbitals of the product is ______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




