Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The root mean square speed of molecules of nitrogen gas at 27$$^\circ$$C is approximately: (Given mass of a nitrogen molecule $$= 4.6 \times 10^{-26}$$ kg and take Boltzmann constant $$k_B = 1.4 \times 10^{-23}$$ J K$$^{-1}$$)
The root mean square speed of gas molecules is:
$$v_{rms} = \sqrt{\frac{3k_BT}{m}}$$
Given that $$T = 27°C = 300$$ K, $$m = 4.6 \times 10^{-26}$$ kg, $$k_B = 1.4 \times 10^{-23}$$ J/K
$$v_{rms} = \sqrt{\frac{3 \times 1.4 \times 10^{-23} \times 300}{4.6 \times 10^{-26}}}$$
$$= \sqrt{\frac{1.26 \times 10^{-20}}{4.6 \times 10^{-26}}}$$
$$= \sqrt{2.739 \times 10^5}$$
$$\approx 523 \text{ m/s}$$
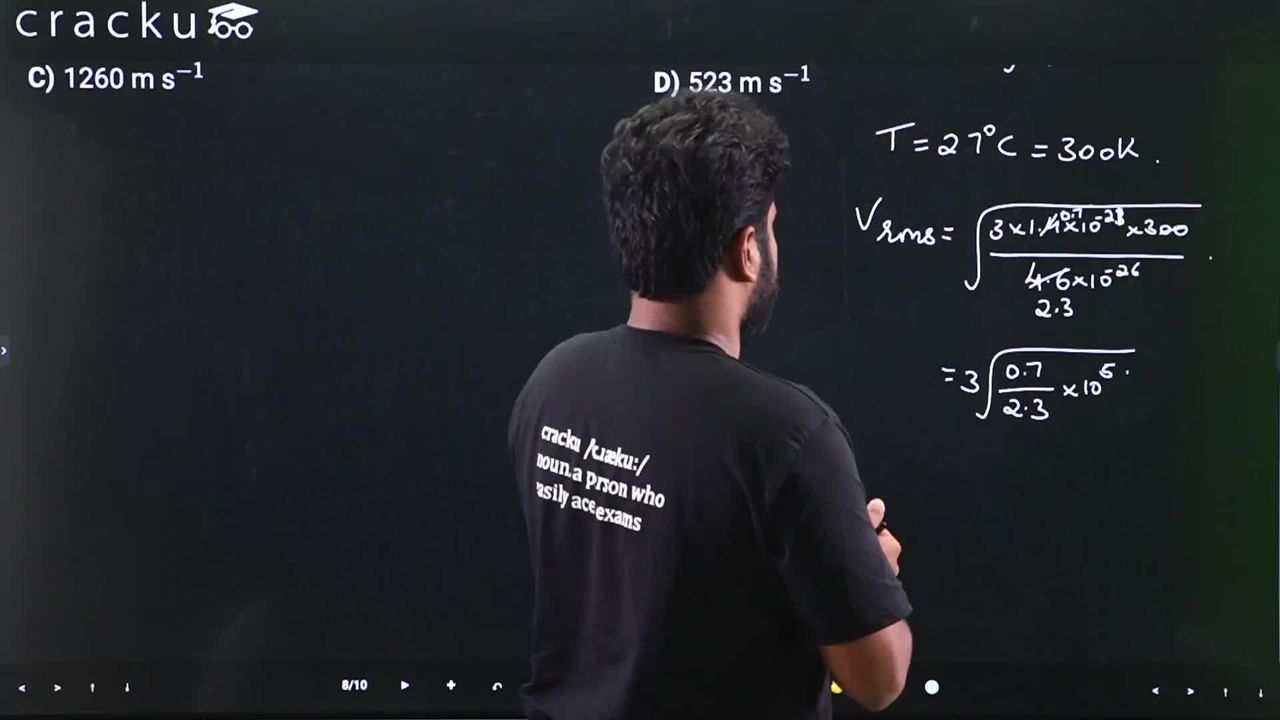
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
