Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A monoatomic ideal gas, initially at temperature $$T_1$$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature $$T_2$$ by releasing the piston suddenly. If $$l_1$$ and $$l_2$$ are the lengths of the gas column, before and after the expansion respectively, then the value of $$\frac{T_1}{T_2}$$ will be:
We need to determine the expression for the ratio of the initial temperature to the final temperature ($$\frac{T_1}{T_2}$$) when a monoatomic ideal gas undergoes a sudden adiabatic expansion.
From the problem statement:
For a monoatomic ideal gas, the ratio of specific heats ($$\gamma$$) is:
$$\gamma = \frac{5}{3}$$
Assuming the cylinder has a uniform cross-sectional area ($$A$$), the volume ($$V$$) occupied by the gas is directly proportional to the length ($$l$$) of the gas column:
$$V = A \cdot l$$
Therefore, the initial volume is $$V_1 = A \cdot l_1$$ and the final volume is $$V_2 = A \cdot l_2$$.
For an adiabatic process, the relationship between temperature ($$T$$) and volume ($$V$$) is given by the formula:
$$T V^{\gamma - 1} = \text{constant}$$
Applying this condition to the initial and final states of the gas system:
$$T_1 V_1^{\gamma - 1} = T_2 V_2^{\gamma - 1}$$
Rearranging the variables to isolate the required temperature ratio $$\frac{T_1}{T_2}$$:
$$\frac{T_1}{T_2} = \left(\frac{V_2}{V_1}\right)^{\gamma - 1}$$
Substitute the length terms in place of the volumes:
$$\frac{T_1}{T_2} = \left(\frac{A \cdot l_2}{A \cdot l_1}\right)^{\gamma - 1} = \left(\frac{l_2}{l_1}\right)^{\gamma - 1}$$
Substitute the value of $$\gamma = \frac{5}{3}$$ into our exponent factor ($$\gamma - 1$$):
$$\gamma - 1 = \frac{5}{3} - 1 = \frac{2}{3}$$
Plugging this back into the temperature ratio equation yields:
$$\frac{T_1}{T_2} = \left(\frac{l_2}{l_1}\right)^{2/3}$$
The value of the temperature ratio $$\frac{T_1}{T_2}$$ is equal to $$\left(\frac{l_2}{l_1}\right)^{2/3}$$, which corresponds exactly to Option B.
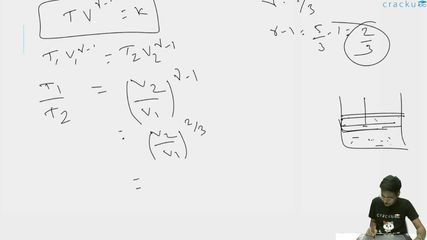
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
