Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following two half-cell reactions along with the standard reduction potential given :
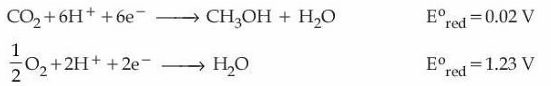
A fuel cell was set up using the above two reactions such that the cell operates under the standard condition of 1 bar pressure and 298 K temperature. The fuel cell works with 80% efficiency. If the work derived from the cell using 1 mol of CH$$_3$$OH is used to compress an ideal gas isothermally against a constant pressure of 1 kPa, then the change in the volume of the gas, $$\Delta V = $$ _____ m$$^3$$. (nearest integer)
Given: $$F = 96500$$ C mol$$^{-1}$$
Correct Answer: 560
The two half-cell reactions with their standard reduction potentials are
$$CO_2 + 6H^+ + 6e^- \to CH_3OH + H_2O,\; E^\circ_{\text{red}} = 0.02\; \text{V}$$
$$\tfrac12 O_2 + 2H^+ + 2e^- \to H_2O,\; E^\circ_{\text{red}} = 1.23\; \text{V}$$
Step 1 Identify cathode and anode.
$$\text{}$$
The higher $$E^\circ_{\text{red}}$$ corresponds to reduction at the cathode.
Hence,
Cathode (reduction): $$\tfrac12 O_2 + 2H^+ + 2e^- \to H_2O$$
$$\text{}$$
Anode (oxidation): reverse the first reaction
$$CH_3OH + H_2O \to CO_2 + 6H^+ + 6e^-$$
Step 2 Balance the electrons.
$$\text{}$$
Multiply the oxygen half-reaction by 3 so both halves involve 6 electrons:
$$\text{}$$
$$\tfrac32 O_2 + 6H^+ + 6e^- \to 3H_2O$$
Step 3 Add the half-reactions.
$$\text{}$$|
$$CH_3OH + H_2O + \tfrac32 O_2 \to CO_2 + 6H^+ + 6e^- + 3H_2O$$
$$\text{}$$
Cancel $$6H^+$$ and one $$H_2O$$ from both sides:
$$\text{}$$
Overall: $$CH_3OH + \tfrac32 O_2 \to CO_2 + 2H_2O$$
Step 4 Calculate the standard cell emf.
$$\text{}$$
$$E^\circ_{\text{cell}} = E^\circ_{\text{cathode}} - E^\circ_{\text{anode}}$$
$$\text{}$$
$$E^\circ_{\text{cell}} = 1.23\; \text{V} - 0.02\; \text{V} = 1.21\; \text{V}$$
$$\text{}$$
Step 5 Find the maximum electrical work for 1 mol of $$CH_3OH$$.
$$\text{}$$
Number of electrons transferred per mole of fuel: $$n = 6$$.
$$\text{}$$
Formula: $$W_{\text{max}} = nF E^\circ_{\text{cell}}$$
$$\text{}$$
$$nF = 6 \times 96500 = 579000\; \text{C mol}^{-1}$$
$$\text{}$$
$$W_{\text{max}} = 579000 \times 1.21 = 700590\; \text{J}$$
$$\text{}$$
Step 6 Account for 80 % efficiency.
$$\text{}$$
$$W_{\text{actual}} = 0.80 \times 700590 = 560472\; \text{J}$$
$$\text{}$$
Step 7 Convert this work into volume change at constant external pressure.
$$\text{}$$
For isothermal compression against a constant pressure, the work needed is
$$\text{}$$
$$W = P_{\text{ext}}\,\Delta V$$
$$\text{}$$
Given $$P_{\text{ext}} = 1\; \text{kPa} = 1000\; \text{Pa}$$, so
$$\text{}$$
$$\Delta V = \frac{W_{\text{actual}}}{P_{\text{ext}}} = \frac{560472}{1000} \approx 560.472\; \text{m}^3$$
$$\text{}$$
Nearest integer: $$\Delta V \approx 560\; \text{m}^3$$.
Create a FREE account and get:
Educational materials for JEE preparation
