Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the three aromatic molecules (P, Q and R) whose structures have been given below :
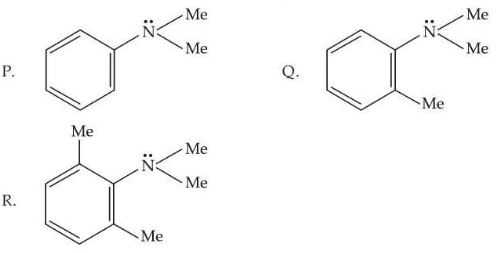
The correct order regarding the reactivity of these compounds with
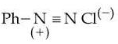
under optimum but slightly acidic medium is :
Azo-coupling with a diazonium ion $$Ph{-}N_2^+Cl^-$$ takes place at those aromatic rings which possess high electron density at the ortho / para positions.
Greater the donating power of the substituent present on the ring under the reaction conditions, faster is the coupling.
The medium recommended in the question is “optimum but slightly acidic”.
Hence we must check whether the -NH2, -NMe2 and -OH groups present in compounds P, Q and R remain in their neutral (electron-donating) or protonated (electron-withdrawing) forms in such a medium.
Compound P : aniline ring (-NH2)
• pKa of anilinium ion $$\left(Ph{-}NH_3^+\right)$$ is about 4.6.
• Even in a mildly acidic solution, most molecules of P are protonated to $$Ph{-}NH_3^+$$, which is a strong meta-directing, electron-withdrawing group.
⇒ Electron density on the ring drops sharply ⇒ lowest coupling rate.
Compound Q : phenol ring (-OH)
• pKa of phenol is ≈ 10.
• In only slightly acidic medium, phenol remains largely in its neutral form $$Ph{-}OH$$ (not as $$Ph{-}OH_2^+$$).
• -OH in its neutral form exhibits a strong +M effect and activates the ring, although not as strongly as an unprotonated amino group.
⇒ Coupling rate is moderate.
Compound R : N,N-dimethylaniline ring (-NMe2)
• The lone pair on nitrogen is strongly released to the ring (+M) while two alkyl groups push electron density through +I effect.
• Protonation constant of a tertiary anilinium ion $$Ph{-}NMe_2H^+$$ is much lower than that of aniline because the electron-releasing Me groups stabilise the neutral base.
• Therefore, in slightly acidic medium, R remains mostly unprotonated and retains its very strong +M effect.
⇒ Highest electron density on ring ⇒ fastest coupling.
Putting the relative activation in order:
$$R \;(Ph{-}NMe_2) \; \gt \; Q \;(Ph{-}OH) \; \gt \; P \;(Ph{-}NH_2$$ or $$Ph{-}NH_3^+)$$
Hence the reactivity sequence towards azo-coupling with $$Ph{-}N_2^+Cl^-$$ is
$$R \; \gt \; Q \; \gt \; P$$
Option C which is: R > Q > P
Create a FREE account and get:
Educational materials for JEE preparation
