Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II.
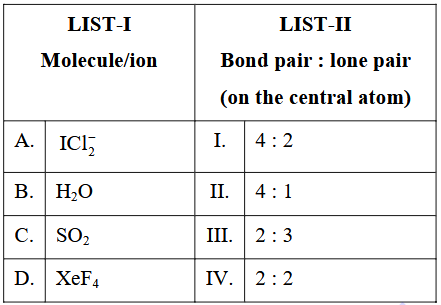
Choose the correct answer from the options given below :
To match each species with the correct “bond-pair : lone-pair” ratio on the central atom we will
(i) count the total valence electrons,
(ii) distribute them around the central atom,
(iii) classify each shared pair as a bond pair (BP) and every remaining pair on the central atom as a lone pair (LP).
Note : When a double bond is present, it contains two shared pairs, i.e. it contributes 2 BP.
Case A: $$ICl_2^-$$
• Iodine has 7 valence electrons, and the anionic charge adds 1 more ⇒ total $$= 8$$.
• Two single bonds to chlorine use 2 pairs ⇒ 2 BP.
• Electrons left on I $$= 8-2 = 6$$, that is 3 lone pairs ⇒ 3 LP.
Hence $$\text{BP : LP} = 2:3$$.
Case B: $$H_2O$$
• Oxygen has 6 valence electrons.
• Two O-H single bonds use 2 pairs ⇒ 2 BP.
• Electrons left on O $$= 6-2 = 4$$, i.e. 2 lone pairs ⇒ 2 LP.
Hence $$\text{BP : LP} = 2:2$$.
Case C: $$SO_2$$
• Sulphur has 6 valence electrons.
• It forms two S=O double bonds.
Each double bond contains two shared pairs, so total bond pairs on S $$= 2 \times 2 = 4$$.
• Electrons left on S $$= 6-4 = 2$$, i.e. 1 lone pair ⇒ 1 LP.
Hence $$\text{BP : LP} = 4:1$$.
Case D: $$XeF_4$$
• Xenon has 8 valence electrons.
• Four Xe-F single bonds use 4 pairs ⇒ 4 BP.
• Electrons left on Xe $$= 8-4 = 4$$, i.e. 2 lone pairs ⇒ 2 LP.
Hence $$\text{BP : LP} = 4:2$$.
Now match with LIST-II:
A. $$ICl_2^-$$ → 2:3 (III)
B. $$H_2O$$ → 2:2 (IV)
C. $$SO_2$$ → 4:1 (II)
D. $$XeF_4$$ → 4:2 (I)
Therefore the correct correspondence is:
A-III, B-IV, C-II, D-I ⇒ Option B.
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.