Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II.
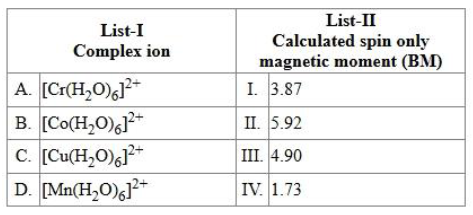
choose the correct answer from the options given below
We first copy the two lists exactly as given in the paper.
LIST-I (Complex) LIST-II (Number of unpaired electrons)
A. $$\left[Fe(H_2O)_6\right]^{2+}$$ I. 3
B. $$\left[Cr(H_2O)_6\right]^{3+}$$ II. 1
C. $$\left[Fe(CN)_6\right]^{4-}$$ III. 4
D. $$\left[Cu(NH_3)_4\right]^{2+}$$ IV. 0
To match each complex with its correct entry in LIST-II we must:
(i) Find the oxidation state of the central metal ion.
(ii) Write the electronic configuration of that oxidation state in the ground state (gas-phase).
(iii) Decide whether the ligand present is weak-field or strong-field. That tells us whether the complex is high-spin or low-spin.
(iv) Fill electrons into the crystal-field or molecular-orbital diagram and count the unpaired electrons.
Complex $$\left[Fe(H_2O)_6\right]^{2+}$$
Oxidation state: $$x + 6(0) = +2 \;\Rightarrow\; x = +2$$, so the metal is $$Fe^{2+}$$.
Gas-phase configuration of $$Fe$$ is $$[Ar]\,3d^{6}4s^{2}$$. Removing two electrons gives $$Fe^{2+}: [Ar]\,3d^{6}$$ (i.e. $$d^{6}$$).
$$H_2O$$ is a weak-field ligand, so the complex is high-spin octahedral. In the high-spin octahedral crystal field the splitting is $$t_{2g}^{4}e_g^{2}$$ with four unpaired electrons.
Hence A matches III (4 unpaired electrons).
Case B:Complex $$\left[Cr(H_2O)_6\right]^{3+}$$
Oxidation state: $$x + 6(0) = +3 \;\Rightarrow\; x = +3$$, so the metal is $$Cr^{3+}$$.
Ground configuration of $$Cr$$ is $$[Ar]\,3d^{5}4s^{1}$$. Removing three electrons gives $$Cr^{3+}: [Ar]\,3d^{3}$$ (i.e. $$d^{3}$$).
Again $$H_2O$$ is weak-field, but for a $$d^{3}$$ ion the splitting pattern is the same in high-spin and low-spin: $$t_{2g}^{3}e_g^{0}$$. That contains three unpaired electrons.
Hence B matches I (3 unpaired electrons).
Case C:Complex $$\left[Fe(CN)_6\right]^{4-}$$
Oxidation state: $$x + 6(-1) = -4 \;\Rightarrow\; x = +2$$, so again the metal is $$Fe^{2+}$$ ($$d^{6}$$).
$$CN^-$$ is a very strong-field (π-acceptor) ligand, so the complex is low-spin octahedral. Electron filling is $$t_{2g}^{6}e_g^{0}$$ with zero unpaired electrons.
Hence C matches IV (0 unpaired electrons).
Case D:Complex $$\left[Cu(NH_3)_4\right]^{2+}$$ (tetra-ammine-copper(II))
Oxidation state: $$x + 4(0) = +2 \;\Rightarrow\; x = +2$$, so the metal is $$Cu^{2+}$$.
Ground configuration of $$Cu$$ is $$[Ar]\,3d^{10}4s^{1}$$. Removing two electrons gives $$Cu^{2+}: [Ar]\,3d^{9}$$ (i.e. $$d^{9}$$).
The complex is square-planar (commonly written with coordination number 4). In both square-planar and octahedral fields a $$d^{9}$$ ion ends up with exactly one unpaired electron (Jahn-Teller distortion in octahedral, or direct splitting in $$D_{4h}$$ symmetry).
Hence D matches II (1 unpaired electron).
Collecting all four matches:
A → III, B → I, C → IV, D → II
This correspondence is exactly Option D.
Final Answer: Option D which is: A-III, B-I, C-IV, D-II
Create a FREE account and get:
Educational materials for JEE preparation
