Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The descending order of basicity of following amines is :
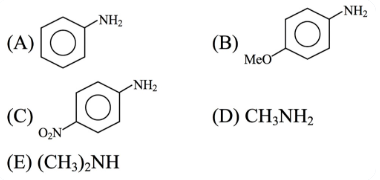
Choose the correct answer from the options given below :
The basicity of an amine depends on how easily the lone pair on the nitrogen can accept a proton. Greater electron density on N and better stabilization of the resulting ammonium ion both increase basicity.
Important effects that decide electron density on nitrogen are:
(i) Inductive effect ($$+I$$ or $$-I$$) from alkyl/aryl substituents.
(ii) Resonance (mesomeric) effect ($$+M$$ or $$-M$$) that may delocalize or donate electron density.
(iii) Hybridisation of the nitrogen (sp$$^3$$ in aliphatic > sp$$^2$$ in aromatic for basicity).
Case 1: Aliphatic amines
$$CH_3NH_2$$ (D) is a primary aliphatic amine.
$$(CH_3)_2NH$$ (E) is a secondary aliphatic amine.
Alkyl groups exhibit a $$+I$$ effect. Two alkyl groups in E push more electron density toward N than one alkyl group in D. In aqueous medium, secondary > primary for basicity (tertiary suffers from poor solvation).
Hence $$\text{basicity: } E \gt D$$.
Case 2: Aromatic amines (anilines)
In aniline, the lone pair on N is partly delocalised into the benzene ring by resonance, making it less available for protonation; therefore aniline is less basic than aliphatic amines.
(A) Aniline - no extra substituent.
(B) p-MeO-Aniline - $$\text{MeO}$$ group shows a strong $$+M$$ (electron-donating) effect, increasing electron density on the ring and on N through resonance, so it is more basic than aniline.
(C) p-NO$$_2$$-Aniline - $$NO_2$$ group shows a powerful $$-M$$ (electron-withdrawing) effect, withdrawing electron density from the ring and N, thus making the amine much less basic.
Therefore, among the three anilines: $$\text{basicity: } B \gt A \gt C$$.
Combining both sets
Overall descending basicity order becomes:
$$(CH_3)_2NH \;(E) \gt CH_3NH_2 \;(D) \gt p\text{-}MeO\text{-Aniline} \;(B) \gt \text{Aniline} \;(A) \gt p\text{-}NO_2\text{-Aniline} \;(C)$$
This matches Option B.
Answer: Option B (E > D > B > A > C).
Create a FREE account and get:
Educational materials for JEE preparation
