Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the reaction $$aX \to bY$$, for which the rate constant at 30°C is $$1 \times 10^{-3}$$ mol$$^{-1}$$ L s$$^{-1}$$. Which of the following statements are true?
A. When concentration of 'X' is increased to four times, the rate of reaction becomes 16 times.
B. The reaction is a second order reaction.
C. The half-life period is independent of the concentration of X.
D. Decomposition of $$N_2O_5$$ is an example of the above reaction.
E.
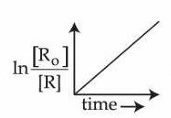
is valid for the above reaction.
Choose the correct answer from the option given below:
The rate law for a reaction is written as $$\text{rate}=k\,[\text{X}]^{n}$$, where $$n$$ is the overall order.
$$\text{}$$
Units of $$k$$ depend on $$n$$ and are given by
$$k=\dfrac{\text{mol}^{1-n}\,\text{L}^{n-1}}{\text{s}}$$.
$$\text{}$$
For the given reaction at 30 °C, $$k=1\times10^{-3}\,\text{mol}^{-1}\,\text{L\,s}^{-1}$$.
$$\text{}$$
Compare this with the general unit expression:
• If $$n=2$$, then $$k=\dfrac{\text{L mol}^{-1}}{\text{s}}=\text{mol}^{-1}\,\text{L\,s}^{-1}$$, which matches the given unit.
$$\text{}$$
• Therefore the reaction is second order with respect to X, i.e. $$n=2$$ and $$\text{rate}=k\,[\text{X}]^{2}$$.
$$\text{}$$
Now test each statement:
Statement A: Increase $$[\text{X}]$$ fourfold: $$\text{rate}\propto [\text{X}]^{2}\Rightarrow (4[\text{X}])^{2}=16[\text{X}]^{2}$$. Rate becomes 16 times ⟹ TRUE.
Statement B: Reaction is second order. We have already established $$n=2$$ ⟹ TRUE.
Statement C: Half-life for a second-order (single-reactant) reaction is $$t_{1/2}=\dfrac{1}{k[\text{X}]_0}$$, which clearly depends on the initial concentration $$[\text{X}]_0$$. Hence it is not independent ⟹ FALSE.
Statement D: Decomposition of $$N_2O_5$$ in the gas phase and in many solvents follows first-order kinetics, not second order ⟹ FALSE.
Statement E: A linear plot of $$\ln\dfrac{[R_0]}{[R]}$$ versus time is the integrated form of a first-order reaction.
For a second-order reaction (single reactant) the linear plot is $$\dfrac{1}{[R]}$$ versus time.
Hence this statement is FALSE.
$$\text{}$$
Only Statements A and B are correct.
$$\text{}$$
Correct option: Option A which is: A and B Only
Create a FREE account and get:
Educational materials for JEE preparation
