Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the reaction 2NO$$_2$$(g) $$\rightleftharpoons$$ N$$_2$$O$$_4$$(g), when $$\Delta S = -176.0$$ J K$$^{-1}$$ and $$\Delta H = -57.8$$ kJ mol$$^{-1}$$ the magnitude of $$\Delta G$$ at 298 K for the reaction is _________ kJ mol$$^{-1}$$. (Nearest integer)
Correct Answer: 5
The standard Gibbs energy change for a reaction is obtained from the relation
$$\Delta G = \Delta H - T\Delta S$$
Given data for the reaction $$2\text{NO}_2(g) \rightleftharpoons \text{N}_2\text{O}_4(g)$$ at $$T = 298\ \text{K}$$:
$$\Delta H = -57.8\ \text{kJ mol}^{-1}$$
$$\Delta S = -176.0\ \text{J K}^{-1}\text{ mol}^{-1}$$
Step 1 - Convert $$\Delta S$$ to the same energy unit as $$\Delta H$$.
Since $$1\ \text{kJ} = 1000\ \text{J}$$,
$$\Delta S = -176.0\ \text{J K}^{-1}\text{ mol}^{-1} = -\dfrac{176.0}{1000}\ \text{kJ K}^{-1}\text{ mol}^{-1} = -0.176\ \text{kJ K}^{-1}\text{ mol}^{-1}$$
Step 2 - Evaluate $$T\Delta S$$ at 298 K.
$$T\Delta S = 298\ \text{K} \times (-0.176\ \text{kJ K}^{-1}\text{ mol}^{-1})$$
$$T\Delta S = -52.448\ \text{kJ mol}^{-1}$$
Step 3 - Substitute in $$\Delta G = \Delta H - T\Delta S$$.
$$\Delta G = (-57.8\ \text{kJ mol}^{-1}) - (-52.448\ \text{kJ mol}^{-1})$$
$$\Delta G = -57.8\ \text{kJ mol}^{-1} + 52.448\ \text{kJ mol}^{-1}$$
$$\Delta G = -5.352\ \text{kJ mol}^{-1}$$
The question asks for the magnitude of $$\Delta G$$. The magnitude is the absolute value:
$$|\Delta G| = 5.352\ \text{kJ mol}^{-1} \approx 5\ \text{kJ mol}^{-1}$$ (nearest integer).
Final Answer: 5 kJ mol−1
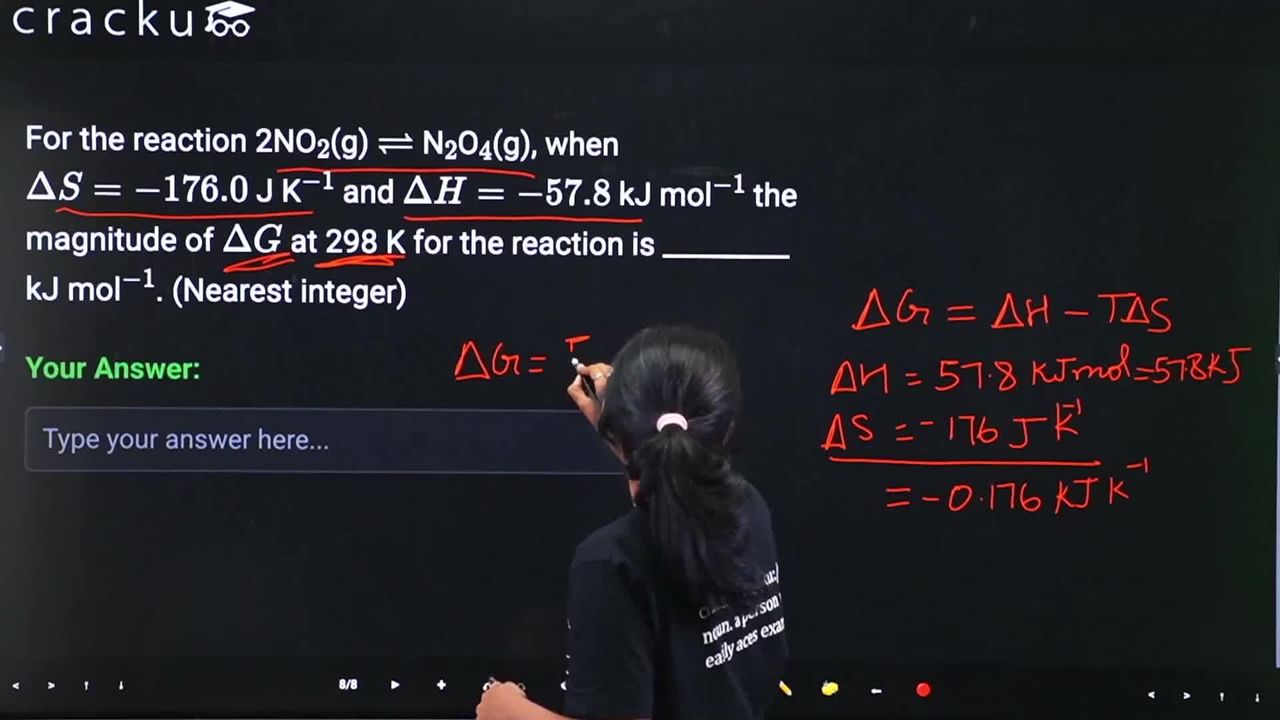
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
